Follow along: IG @birectifier
Kervegant Chapter III Distillery Fermentations: [PDF]
Pages 67-93
CHAPTER III Distillery Fermentations (1)
(1) Van Lear – La chimie des fermentations. Paris 1935.
Sweet musts left to themselves are the seat of complex phenomena. Parallel to or after the alcoholic fermentation, secondary fermentations (acetic, butyric, lactic, putrid, etc.) develop. These always have the effect of reducing alcohol yield. So they are considered undesirable and usually distillers do their best to avoid them, especially in an industrial alcohol distillery. In certain cases, however, when one wishes to obtain eaux-de-vie with pronounced bouquet, secondary fermentations generating aromatic principles are allowed and even promoted. In the rhummerie, the importance of the latter is most variable. In the production of light rums, pure yeasts and sterilized or antiseptic media are used to achieve pure alcoholic fermentations. On the other hand, microbial agents, and mainly butyric bacteria, play a very important role in the production of Jamaica’s very heavy rums. Between these extremes, we find all the intermediate fermentation complexes.
Alcoholic fermentation.
It is only at the end of the eighteenth century that the first precise information on alcoholic fermentation dates back. Lavoisier, applying his methods of analysis, showed that sugar was splitting into alcohol and carbon dioxide. Few years later, Gay-Lussac translated these results into the general formula:
C6H1206 = 2 C2H6O+ 2 CO2
Lavoisier had only considered the chemical aspect of the phenomenon, without expressing any hypothesis as to the nature or the mode of action of the ferment indispensable to effect the transformation. Although this ferment was observed under the microscope and described by Leuwenhoek in 1680, its nature as a living being was established only around 1825, by Turpin and Cagnard-Latour in France, Schwann and Kutzing in Germany. Pasteur showed, in 1859, that fermentation was a correlative process of yeast life, contrary to Liebig’s theory that the decomposition of sugar was due to a molecular shattering caused by fermentation. While recognizing the living nature of yeast, various scientists, including Claude Bernard and Berthelot, believed that its action on sugar was due to a diastase separable from the cell. The Buchner brothers were able to isolate, in 1897, the alcoholic zymase. Harden and Young have shown that it is formed by a mixture of two substances, inactive separately and recovering their activity when they are mixed: one colloidal and losing its fermentative properties by heating, apozymase or ferment itself; the other dialysable and thermostable, the cozymase or coferment of the zymase. [SOS something seems amiss in the last lines here.]
Mechanism of fermentation.
The classic Gay-Lussac formula gives only a very general idea of the complex phenomenon of alcoholic fermentation. It does not take into account intermediate bodies or secondary products (succinic acid, glycerin, etc.) which form. It is now generally agreed that carbonic acid and alcohol derive from a 3-carbon body, which itself derives from the breaking up of the hexose molecule. Bayer (1870), and after him Wohl, regarded as an intermediate body lactic acid, which is often found among the products of alcoholic fermentation. In a first phase, zymase would transform glucose into lactic acid, while in a second phase another diastase, lactacidase, break down this acid into ethyl alcohol and carbon dioxide. In fact, if lactic acid could be obtained by reacting yeast juice with glucose solutions (Oppenheimer), it was not possible to convert lactic acid into alcohol by live yeast. So this theory is abandoned today. We then thought of dioxyacetone CH2OH – CO – CH2OH (Wohl, Buchner, Fernbach). This could be obtained chemically and biologically at the expense of hexoses and gives alcohol by fermentation. Unfortunately, it has so far been tried in vain to produce it by yeast, so that its formation during the alcoholic fermentation remains purely hypothetical. According to other authors, notably Neuberg, the first step in the cleavage of the hexose molecule would result in hydrated methylglyoxal (or pyruvic aldehyde). This one, by simple dehydrogenation, would give the pyruvic acid, which would decompose, under the action of a carboxylase, in acetaldehyde and carbon dioxide. Finally, acetaldehyde would provide, by reduction, ethyl alcohol. These successive transformations are summarized by the following equations (Neuberg scheme):  The formation of pyruvic acid during alcoholic fermentation was demonstrated by Fernbach and Schoen, who were able to obtain it by adding chalk to the liquid, so as to prevent any acidification of the medium. This acid decomposes very easily under the influence of yeast to give acetaldehyde and carbon dioxide. Acetaldehyde has also been obtained directly in fermentations carried out by living yeast: by adding alkali sulphites to the must, it is easy to bind (Müller-Thurgau, Neuberg). Finally acetaldehyde can be reduced by yeast, giving alcohol (Kostytschew). There is therefore a growing tendency to admit that pyruvic acid and acetaldehyde are indeed intermediate products between sugar and alcohol. With regard to pyruvic aldehyde, the question is more debated. The presence of methylglyoxal has only been observed in a small number of cases, especially in bacterial fermentations (Fernbach, Aubel). It is only under special conditions, in the absence of coferment, that Neuberg observed his formation from glucose, under the action of yeast extracts. In addition, methylglyoxal is not fermentable by yeast (1). It is therefore considered today, with Meyerhoff, that this aldehyde is an accidental product of fermentation, arising purely by chemical means, and that the precursor of pyruvic acid is probably glyceric acid. (1) This can not, however, be considered as absolute proof. It has been pointed out, in fact, that the form in which methylglyoxal takes place during sugar metabolism may be much more labile than in the state in which we handle it. In addition, the product, which is normally used as it is formed without ever accumulating in the medium, risks, if introduced in bulk, to distort the mechanism of transformations. These arguments, which also apply to other intermediate products, have often been used by authors to explain the difficulties of fermenting these substances in vitro. It is generally admitted, following the work of Harden and Young, that phosphoric acid combines with sugar, in the presence of zymase, to give a phosphorus-containing organic compound, hexose-diphosphate or fructose-diphosphate, which is attributed to following formula:
The formation of pyruvic acid during alcoholic fermentation was demonstrated by Fernbach and Schoen, who were able to obtain it by adding chalk to the liquid, so as to prevent any acidification of the medium. This acid decomposes very easily under the influence of yeast to give acetaldehyde and carbon dioxide. Acetaldehyde has also been obtained directly in fermentations carried out by living yeast: by adding alkali sulphites to the must, it is easy to bind (Müller-Thurgau, Neuberg). Finally acetaldehyde can be reduced by yeast, giving alcohol (Kostytschew). There is therefore a growing tendency to admit that pyruvic acid and acetaldehyde are indeed intermediate products between sugar and alcohol. With regard to pyruvic aldehyde, the question is more debated. The presence of methylglyoxal has only been observed in a small number of cases, especially in bacterial fermentations (Fernbach, Aubel). It is only under special conditions, in the absence of coferment, that Neuberg observed his formation from glucose, under the action of yeast extracts. In addition, methylglyoxal is not fermentable by yeast (1). It is therefore considered today, with Meyerhoff, that this aldehyde is an accidental product of fermentation, arising purely by chemical means, and that the precursor of pyruvic acid is probably glyceric acid. (1) This can not, however, be considered as absolute proof. It has been pointed out, in fact, that the form in which methylglyoxal takes place during sugar metabolism may be much more labile than in the state in which we handle it. In addition, the product, which is normally used as it is formed without ever accumulating in the medium, risks, if introduced in bulk, to distort the mechanism of transformations. These arguments, which also apply to other intermediate products, have often been used by authors to explain the difficulties of fermenting these substances in vitro. It is generally admitted, following the work of Harden and Young, that phosphoric acid combines with sugar, in the presence of zymase, to give a phosphorus-containing organic compound, hexose-diphosphate or fructose-diphosphate, which is attributed to following formula:  According to Meyerhoff (2). this body would split, under the action of a diastase, zymohexase, in 2 molecules of a triose-monophosphate, phosphoglycérique aldehyde or phosphoglycéral: (2) Ann. Inst. Pasteur LIII, 221, 1934.
According to Meyerhoff (2). this body would split, under the action of a diastase, zymohexase, in 2 molecules of a triose-monophosphate, phosphoglycérique aldehyde or phosphoglycéral: (2) Ann. Inst. Pasteur LIII, 221, 1934. ![]() Two phases should be considered, differing in their speed: the incubation or induction phase, which is slower, and the actual fermentation or stationary phase. The first would involve transformation of the phosphoglyceric aldehyde, by oxidation-reduction, into the corresponding acid and alcohol (phosphoglycerolic acid and phosphoglycerol):
Two phases should be considered, differing in their speed: the incubation or induction phase, which is slower, and the actual fermentation or stationary phase. The first would involve transformation of the phosphoglyceric aldehyde, by oxidation-reduction, into the corresponding acid and alcohol (phosphoglycerolic acid and phosphoglycerol):  Phosphoglycerol (or phosphoglycerol alcohol) stays away from the fermentation cycle (saponified, it gives rise to glycerin), while phosphoglyceric acid breaks down into pyruvic acid and phosphoric acid:
Phosphoglycerol (or phosphoglycerol alcohol) stays away from the fermentation cycle (saponified, it gives rise to glycerin), while phosphoglyceric acid breaks down into pyruvic acid and phosphoric acid: ![]() Pyruvic acid gives, by decarboxylation, acetaldehyde:
Pyruvic acid gives, by decarboxylation, acetaldehyde: ![]() We then go to the stationary phase where the speed of the phenomenon accelerates: acetaldehyde reacts on the phosphoglyceric aldehyde, to form by oxidation-reduction, alcohol and phospoglycérique acid:
We then go to the stationary phase where the speed of the phenomenon accelerates: acetaldehyde reacts on the phosphoglyceric aldehyde, to form by oxidation-reduction, alcohol and phospoglycérique acid:  The phosphoglyceric acid decomposes, according to reactions 3 and 4, to release carbon dioxide, phosphoric acid and acetaldehyde which enters the reaction.
The phosphoglyceric acid decomposes, according to reactions 3 and 4, to release carbon dioxide, phosphoric acid and acetaldehyde which enters the reaction.
In support of Meyerhoff’s theory, the following facts are invoked. By the use of various fermentation paralyzers, it was possible to highlight the above transformations. Thus, monoiodoacetic acid prevents the decomposition of the phosphoglyceric aldehyde and stops the transformation when the reaction (1) has been completed. Fluoride hinders reaction (3): a mixture of phosphoglycerol and phosphoglyceric acid is obtained in its presence. Finally, by adding alkaline sulphites to the must, pyruvic acid and glycerine are formed. It has also been observed that addition of acetaldehyde to a sweet must completely suppresses the period of fermentation induction.
[I think we are missing two foot notes here. page 69]
It remains, however, that hexose diphosphate, if it has been possible to obtain it by action of yeast extracts on fermentable sugars, has not yet been isolated from the musts in fermentation, and that, if introduced in the musts, it is not transformed by the yeast. It is then reduced to suppose that the diphosphoric ether is used as it is produced, and that the one which is added to the liquid does not penetrate inside the yeast cells.
It has also been observed that various phosphoric ethers above (hexose-diphosphate, phosphoglycerol, phosphoglycerol and phosphoglyceric acid) ferment less easily than sugars in the presence of yeast extracts. They may originate in a more labile and fermentable form than that which we know or intervene in the reactions of activators we do not know (1).
(1) In the presence of traces of arseniate, for example, the fermentation of phosphoric intermediates is greatly accelerated.
A nitrogenous base, adenosine, appears to play, in the form of adenylic acid (adenine monosphosphoriboside) and adenosine-phosphoric acid (adenine triphosphoriboside), which is always found in yeast, an essential role in the phosphorylation of sugars.
Adenosine triphosphoric would be the lessor of phosphoric acid for the etherification of sugars, and phosphoric acid, by regenerating the acid adenine-triphosphoric acid. This appears then as the catalytic factor of the phosphorylation and would constitute, according to some authors, the cozymasé.
Secondary products of fermentation.
During alcoholic fermentation, ethyl alcohol and carbon dioxide are also formed, various secondary products which come, some of the “vegetable function” of the yeast (higher alcohols, succinic acid, etc.), others of the “zymatic function” (glycerine, aldehyde acetic, etc.). The nature and proportion of these products vary within wide limits, according to conditions of the medium, yeast being “a plastic organism, whose physiological properties can undergo important modifications under the influence of various factors” (Fernbach).
Glycerin. — In his “Memoir on Alcoholic Fermentation” (1859), Pasteur reported for the first time that glycerine or glycerol, CH2OH – CHOH – CH2OH, was a constant product of fermentation.
Laborde (2) observed that the quantity of glycerine, relative to 100 g of sugar metabolised, varied within wide limits: 2.50 to 7.75 g (2.5 to 3.5 on average). He showed that all the causes of weakening of the yeast contributed to increase the production of glycerine: long fermentation duration, richness of sugar, abundant yeast multiplication by aeration, etc. The nature of sugar and the yeast race also have an influence.
(2) C. R. CXXIX, 344, 1899, Ann. Brass. Dist. II, 473.
Neuberg, then Connstein and Ludecke (3) have shown that, by addition of sodium sulphite to the must, there is an increase in the yield of glycerine, up to 36% of the sugar. This process was used industrially in Germany, during the 1914-18 war, to obtain the glycerin necessary for manufacture of explosives.
(3) Ber. Deut. Chem. Ges. LII, 1385, 1919.
Another method, attributed to Eoff, Linder and Beyer (4), is to ferment sugar in the presence of baking soda (or other alkalis), which is added to the liquid after the start of fermentation, gradually and in small portions up to a dose of 5%. 20 to 25% of the sugars in the must are converted to glycerin, the balance providing alcohol, acetic acid and carbon dioxide, with small amounts of various other poorly identified products.
(4) Ind. Eng. Chem. XI, 843-45, 1919.
Glycerine is generally considered a “byproduct” of processing sugar into alcohol. When the chain of reactions is deflected, preventing the medium from being acidified or adding sulfite, the chemical specificity of the yeast, which becomes a glyceric ferment, is modified. We would have in the first case: ![]() and in the second case:
and in the second case:  In the Neuberg scheme, pyruvic acid or acetaldehyde is fixed by alkalis or sulphite, and hydrogen coming from reduction of methyglyoxal would react with one of the molecules of the latter, which it would transform into glycerin.
In the Neuberg scheme, pyruvic acid or acetaldehyde is fixed by alkalis or sulphite, and hydrogen coming from reduction of methyglyoxal would react with one of the molecules of the latter, which it would transform into glycerin.
According to Meyerhoff’s theory, glycerine would come from saponification of phosphoglycerol, with bisulphite intervening to block ethyl aldehyde [acetaldehyde by another name] as it is formed and prevent its reaction with phosphoglycerol aldehyde.
It is explained that a certain amount of glycerin always occurs in alcoholic fermentation and alcohol in glycerol fermentation, because the yeast never behaves like a pure alcoholic ferment or a pure glyceric ferment. The two transformations would develop side by side, in varying proportions according to conditions of the environment.
In ordinary alcoholic fermentations, glycerin is formed on the whole at the end of the fermentation, when only a small quantity of sugar remains, and nutrition conditions are unfavorable to the yeast.
Aldehydes — Acetic aldehyde, or acetaldehyde, CH3-COH, has long been reported among the products of alcoholic fermentation (Roeser, 1893). It usually exists only in very small quantities. However, in wines, it was possible to isolate up to 860 mgr of aldehyde per 100 grs of sugar consumed.
It is generally accepted that it constitutes an intermediate body in the transformation of sugar into alcohol. Muller-Thurgau and Osterwalder have shown that sulphurous acid and sulphites, added to the musts of fermentation, combine with aldehyde, which is thus fixed and can no longer be converted into alcohol. In this way 18-24% of the sugar could be transformed in the form of aldehyde. Liquids fermented in the presence of SO2 would thus normally contain a larger amount of acetic aldehyde.
Acetaldehyde can also be produced by oxidation of ethyl alcohol. Kayser and Demolon have observed that resting wines on lees, in broad contact with the air, favored production of aldehyde and that the living yeast was the essential agent of this phenomenon. Trillat and Sauton (1) showed that yeasts were likely to produce significant amounts of acetaldehyde at the expense of alcohol within a few hours: up to 2,500 mg per 100 alcohol at 100° after 6 hours. Aldehyde formed disappears little by little, if the contact with the yeast is prolonged.
(1) C. R. CXLVI, 998, 1908.
Finally, aldehydeification of alcohol can take place by purely chemical means, especially during distillation and when the eaux-de-vie are treated with porous (charcoal filtration) or oxidizing (oxygenated water, permanganate, etc.).
Furfurol, or pyromucic aldehyde, mainly arises during distillation, by prolonged boiling or roasting of certain carbohydrates. Kayser and Demolon, however, believe that the yeast itself must intervene in its production. These authors observed that proportion of furfurol underwent variations according to nitrogen diet: it was minimal in the case of yeasts receiving sulphate of ammonia and instead increased substantially in the presence of leucine. It is not impossible, therefore, that furfurol constitutes an excretion product, linked to nitrogen disassimilation.
Higher Alcohols. — It is only towards the end of 19th century, following the works of Pasteur, Wurtz, Windisch, etc… that the composition of the oil of fusel, or “fusel oil”, whose presence had been known since Scheele (1785), could be specified. This consists of a mixture of higher alcohols, qualitatively and quantitatively variable depending on the origin of the eau-de-vie, but among which amyl and butyl alcohols predominate.
Today, these higher alcohols have two different origins. Some (normal butyl alcohol) come from the attack of carbohydrates by bacteria, while others (amyl alcohol, isobutyl alcohol, etc.) result from degradation of amino acids by yeast. High levels of higher alcohols in alcoholic liquids almost always result from bacterial interventions.
F. Ehrlich (1) showed, in 1905, that higher alcohols which originate during alcoholic fermentation are the by-products of the nitrogen nutrition of yeast. When it has at its disposal, as nitrogenous food, only amino acids, it is to the amino group of these latter that it borrows the nitrogen which is necessary for construction of its cells. Amino acids first undergo de-amination (loss of NH3, then decarboxylation (loss of CO2), ultimately to provide an alcohol containing one carbon atom less than the amino acid that gave rise to it. Thus isobutyl alcohol would originate from valine, isoamyl alcohol from leucine:
(1) Z. Ver. Rubenzuckr. Ind. 1905, 539 : Ber. Deut. Chem. Ges. XL, 1027, 1907  Thorme (2), adding different amino acids to a nutrient medium, was able to recover, after fermentation by yeast, the corresponding higher alcohols, in amounts closely proportional to those of the amino acids employed.
Thorme (2), adding different amino acids to a nutrient medium, was able to recover, after fermentation by yeast, the corresponding higher alcohols, in amounts closely proportional to those of the amino acids employed.
(2) J. Inst. Brewing XLII. 13, 1939.
Ehrlich assumed that production of higher alcohols at the expense of amino acids was the result of simple hydrolysis and could be represented by the general formula: ![]() The reaction would actually be much more complex, according to Neubauer and Fromherz (3), who consider that the process of amino acid degradation actually corresponds to an oxidation-reduction, and proceeds as follows:
The reaction would actually be much more complex, according to Neubauer and Fromherz (3), who consider that the process of amino acid degradation actually corresponds to an oxidation-reduction, and proceeds as follows:
(3) Z. Physiol. Chem. LXX, 326, 1910.
1°) oxidation of the amino acid and production of a hydrate of the corresponding amino acid: ![]() 2°) proper de-amination, giving rise to the ketonic acid of the same series:
2°) proper de-amination, giving rise to the ketonic acid of the same series: ![]() 3°) decarboxylation and production of an aldehyde having one less carbon atom than the amino acid:
3°) decarboxylation and production of an aldehyde having one less carbon atom than the amino acid: ![]() 4°) finally reduction and production of the higher alcohol:
4°) finally reduction and production of the higher alcohol: ![]() This process has a striking analogy with that of sugar fermentation: the ketonic acid obtained in the second stage of the transformation is a homologue of pyruvic acid and the last two stages are the same in one and the other case.
This process has a striking analogy with that of sugar fermentation: the ketonic acid obtained in the second stage of the transformation is a homologue of pyruvic acid and the last two stages are the same in one and the other case.
Ammonia from the disaggregation of amino acids can never be found as such in musts. It must therefore be converted into insoluble protein materials, forming part of the yeast cells.
Ehrlich observed that in ordinary fermentations of pure sugar, the amount of fusel oil produced ranged from 0.4 to 0.7% of the alcohol formed. These figures are close to those achieved in industrial practice. Thus Martraire (1) was able to obtain, with apple musts, an average production of 0.512% fusel oil to alcohol in the year 1928 and 0.520% in 1929. Houssiau (2), in 1932 and 1936, in the case of musts of beet and cane molasses, an average annual yield of 0.306 to 0.423% of crude alcohol and 0.191 to 0.265% of sugar was observed (3).
(1) in Pérard. — Le rendement en distillerie [Distillery yield]. C. R. 5. Cong. Int. des Ind. Agr. 802 – 832, 1937.
(2) C. R. 5. Cong. Int. des Ind. Agr. 864, 1937.
(3) The figures given, which correspond to the fusel oil obtained industrially, are somewhat inferior to reality, because part of the higher alcohols (10%) is removed during the distillation with bad tasting tail.
The production of fusel-oil is higher, when fermentation is carried out by yeast low in nitrogen, low fermentative power, working in the presence of a large amount of sugar (the proportion of 1 yeast for 5 of sugar is very favorable). On the other hand, when fermentation takes place rapidly, via a large quantity of yeast rich in nitrogen, decomposition of amino acids is relatively weak (Ehrlich).
The proportion of higher alcohols formed also depends on the nitrogenous matter composition of the must. Yeast uses amino acids only if it does not have another source of nitrogen that is more easily assimilated. When adding asparagine to the must, or better, carbonate or sulphate of ammonia, the production of fusel-oil is very much diminished, even in the presence of large quantities of leucine (Ehrlich, Pringsheim). However, it is not possible to completely suppress this production. Pringsheim (4) found that a dose of 250 gr. Ammonium sulphate per 1,000 liters of must was enough to reduce to a minimum the quantity of higher alcohols formed; by doubling the dose of ammonia salt, the rate of fusel oil remains the same.
(4) Biochem. Z. III, 121, 1907 : X, 490, 1908.
On the other hand, addition of amino acids, and more particularly leucine, to a sugar solution increases fusel oil production, the proportion of which could, according to Ehrlich, be 3% under favorable conditions. The amount of higher alcohols, however, is still limited here, because the yeast uses amino acids only in so far as it needs their nitrogen to build the protein materials used in the constitution of its cells. The excess of amino acids remains unconverted.
Thus, Houssiau observed that the nitrogen of the amino acids disappearing during fermentation (molasses, cane and beet musts) was 30 to 60%, and Martraire that the amount of amino acids used was sensibly constant. (0.050 gr of N per liter), whatever the initial content of the liquid in amino acids.
Under certain conditions, production of higher alcohols could, however, be much greater. Salesskaja (5) obtained with musts containing 10% sugar, largely aerated and with 0.5% leucine added, a fusel-oil yield of 13.5% of the alcohol or 0.16% of the must, which represents an assimilation of 70 to 80% of the amino nitrogen. Increasing the amount of leucine does not cause the increase in the level of higher alcohols. Replacement of leucine by another amino acid lowers the proportion of fusel oil to 0.08 and even 0.04% of must. [I think I translated the logic of that correctly.]
(5) Mikrobiologija VI, 68, VII, 66, 1938.
Yeast does not use only the naturally occurring amino acids in musts, but also those derived from disassimilation of nitrogenous principles and autolysis of old cells. Thus, some authors explain the formation of isobutyl alcohol from valine, a normal product of the degradation of yeast albumins. However, according to Ehrlich, amino acids excreted by yeast would only be of great value if, as a result of defective nitrogen nutrition, high temperature or other adverse conditions, partial autolysis of the old cells occurs.
The composition of the fusel-oil varies within wide limits, according to the nature of the amino acids of musts, the conditions under which fermentation takes place, etc. In general, there is dominance of isoamyl alcohol, which sometimes constitutes up to 80% of the fusel oil, with varying amounts of normal isobutyl and propyl alcohol. Butyl and isopropyl alcohols are the result of bacterial interventions; they can be found in large quantities in alcohols from must left for spontaneous fermentation.
Kayser and Demolon have observed that production of higher alcohols is favored by light and that of volatile acids by darkness. According to these authors, formation of higher alcohols would be related to yeast multiplication intensity. It is, in fact, increased in the presence of high doses of peptone, which promotes cell multiplication and decreased if we add asparagine to the must, which increases the speed of fermentation by reducing the yield of yeast. In other cases, it has been found that the amount of higher alcohols formed varied in the same direction as the weight of yeast recovered.
Jankovic (1) has observed that factors favoring yeast activity also increase fusel oil production. Having fermented a molasses must (which contained 52.4% sugars and 1.7% nitrogen) under different conditions of concentration and temperature, he found that the maximum amount of fusel oil (1.45%) was obtained when the density of the must was 15° Brix, the acidity 0.5 and temperature 27°; and the minimum amount (0.22%), if the density was raised to 25° Brix and the acidity to 1.5, temperature remaining at 27° C.
(1) Z. Spiritusind. LI, 106, 1928.
Different races of yeast seem to be rather little differentiated in the production of the fusel oil, according to Kayser and Demolon. However, Schizosaccharomyces gives lower amounts of higher alcohols than budding yeasts (Kayser). On the other hand, Arroyo found notable differences in the production of higher alcohols, according to yeast races. He obtained the following results, using cane juice as a culture medium and operating at a temperature of 33-35°. 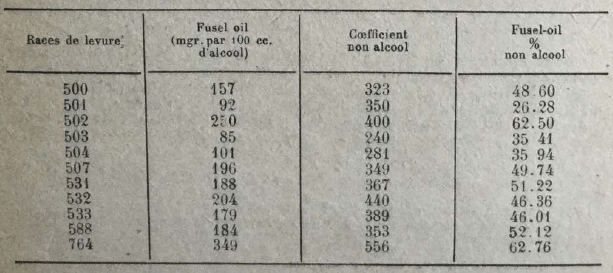 The same author has also observed that formation of higher alcohols diminishes when fermentation takes place at relatively low temperatures and when yeast is fed with liquid ammonia in place of sulphate of ammonia. Hereinafter, the amounts of higher alcohols obtained in various tests (in grams per hl. alcohol at 100°).
The same author has also observed that formation of higher alcohols diminishes when fermentation takes place at relatively low temperatures and when yeast is fed with liquid ammonia in place of sulphate of ammonia. Hereinafter, the amounts of higher alcohols obtained in various tests (in grams per hl. alcohol at 100°).  Methyl alcohol [Methanol]. — As a result of the works of Fellenberg, Buchka, Takahashi, Flanzy, etc., we tend to admit that methyl alcohol is found in variable quantities, but generally low, in all liquids which have undergone alcoholic fermentation.
Methyl alcohol [Methanol]. — As a result of the works of Fellenberg, Buchka, Takahashi, Flanzy, etc., we tend to admit that methyl alcohol is found in variable quantities, but generally low, in all liquids which have undergone alcoholic fermentation.
Von Fellenberg (1) showed that pectins could be considered as methyl esters of pectic acids and contained on average 10% of methyl alcohol. Under influence of various causes (heating with dilute acids, alcoholic fermentation, etc.), pectic substances are decomposed, with release of methyl alcohol. Pectoses and lignins can also give birth to this alcohol, but with more difficulty than pectins.
(1) Mitt. Lebennsm. Hyg. V, 172, 1914.
According to Flanzy, during alcoholic fermentation, and in the absence of any pectic product, a certain quantity of methyl alcohol, variable with the nature of the sugar, would be produced: glucose, for example, would give more methyl alcohol than levulose.
It would be especially at the beginning of the fermentation that methyl alcohol would appear, at the same time that pectic compounds undergo a profound modification resulting in their de-methylation and liberation of their constitutive phosphorus (Flanzy).
Acids. — The acidity of fermented liquids is due to fixed (lactic acid, succinic acid, etc.) or volatile acids (formic acid, acetic acid, butyric acid, etc.) brought by the raw materials of the must or produced during fermentation.
According to Arroyo, the quantity of acids formed by yeast during fermentation is usually the equivalent of 4.7 to 10.0 cc of decinormal soda per 100 cc of must. Volatile acids correspond to 2.1-5.8 and acids to 2.1-5.4 cc decinormal soda.
[This is the baseline that the Δ acidity concept is abased on. Decinormal can also be said as 0.1 N]
The presence of succinic acid, CO2H-H2-CH2-CO2H, among the products of alcoholic fermentation has been reported for the first time by Pasteur. The amount of this acid (usually 0.5 to 0.8% of processed sugar) is very variable. It depends mainly on yeast race and original acidity of the must (Ventre).
Pasteur thought that succinic acid resulted from sugar processing. It is admitted today, following the works of Ehrlich and Neuberg, that it comes from decomposition of proteic matters, under the influence of yeasts or putrefactive microbes. It appears to derive mainly from glutamic acid, by a process analogous to that of the formation of higher alcohols from amino acids: ![]() While succinic acid appears at the end of fermentation, volatile acids are formed during early stages of fermentation (Reisch, Oster walder); their production ceases practically when the amount of sugar has been reduced to 50% (Joslyn and Dunn). However, when the fermentation is finished, they increase again, as a result of the oxidation of the alcohol. Osterwalder (1), confirming the results of Kayser and Demolon, showed that yeasts preserved in the fermented must could produce as much as 1.8 per 1,000 of volatile acids.
While succinic acid appears at the end of fermentation, volatile acids are formed during early stages of fermentation (Reisch, Oster walder); their production ceases practically when the amount of sugar has been reduced to 50% (Joslyn and Dunn). However, when the fermentation is finished, they increase again, as a result of the oxidation of the alcohol. Osterwalder (1), confirming the results of Kayser and Demolon, showed that yeasts preserved in the fermented must could produce as much as 1.8 per 1,000 of volatile acids.
(1) Cent. Bakt. Abt. II, XXXII, 481.
The quantity and nature of volatile acids varies not only with various factors which act on the development of the ferments (reaction of the medium, temperature, aeration), but again, especially with yeast races.
Fernbach (2) has shown that the total acidity formed during fermentation was all the greater when the liquid was originally less acidic: in the tests carried out, it varied from 59.5 to 123.5 mgr per liter (tartaric acid), when the original must was neutral.
[I think neutrality here is a pH concept. This follows Arroyos advice to create a large buffer and start the fermentation of a full flavored rum higher in pH than a light rum.]
(2) C. R. CLVI, 77, 113.
Ventre (3) observed in the case of neutral grape musts, a fixed acid production of 1-1.7% of sugar consumed, each yeast having a particular way of forming fixed acidity and volatile acidity. In acidic environments, however, there may be a decrease in acidity during fermentation, as a result of the yeast attack of certain organic acids (primarily malic acids) in the wort.
[This may be a paper worth seeking out to see if it refers to S. Pombe yeasts metabolizing malic acid.]
(3) C. R. CLVII, 154, 1913.
Kayser found that the quantity and also nature of volatile acids produced depended to a large extent on yeast race. Film yeasts, for example, give rise to large quantities of acids, with notable production of formic acid. Nitrogen diet also has a significant influence, but varies according to yeast races in the ferment.
According to the same author, the main volatile acids which originate in the fermentation of cane molasses by pure yeasts are acetic, butyric and formic acids. The first of these acids is in general largely predominant and sometimes alone accounts for almost all of the volatile acidity. Formic acid, however, would be produced in appreciable quantity by certain yeasts (film yeasts) and under certain conditions of nitrogen supply (nitrogen amide). According to Thomas (4), if yeast is cultivated in the presence of acetamide or time, formic acid is formed almost exclusively.
(4) Ann. Inst. Pasteur XXXIV, 162, 1920.
Srinivasan found in fermented cane molasses, besides formic, acetic and butyric acids, propionic acid in noticeable quantities; and various authors have reported the presence, in fermented liquids and eaux-de-vie, of high molecular weight fatty acids: caproic, capric, lauric, palmitic, etc.
It is now admitted that acetic acid is a byproduct of sugar conversion into alcohol and that it results from the oxidation of acetic aldehyde (Kostyjschew, Neuberg). It can also come from the oxidation of ethyl alcohol by yeast after fermentation (Kayser and Demolon, Osterwalder). However in practice, the presence of significant amounts of this acid is always related to the intervention of acetic bacteria, parallel or subsequent to the alcoholic fermentation.
According to Thomas, formic acid results from protoplasmic yeast activity, oriented in a particular direction by the supplied nitrogenous foods. It may also derive, at least in the case of molasses musts, directly from sugar by a purely chemical route (Wilson).
According to a widespread opinion. the other acids which are formed during alcoholic fermentation would come from the dis-assimilation of the yeast cells. Their formation was attributed by Duclaux to the mechanism of nitrogen nutrition. Thus, ketone acids from the conversion of amino acids, instead of reducing the higher alcohols, can, under certain conditions, oxidize acids having fewer carbon atoms than amino acid from which they come: ![]() Hydrolysis of certain amides also sometimes gives rise directly to acids:
Hydrolysis of certain amides also sometimes gives rise directly to acids: ![]() E. Luce (1) admits that acids also come, at least partly, from yeast fats. These would first be split into glycerin and fatty acids, which would then be degraded, as in Knop’s ß oxidation theory.
E. Luce (1) admits that acids also come, at least partly, from yeast fats. These would first be split into glycerin and fatty acids, which would then be degraded, as in Knop’s ß oxidation theory.
(1) J. Pharm, Chim, (7), XXII, 138, 1920.
Under influence of oxidants, normal acyclic acids are converted into oxacids, corresponding to β-, that is to say on the penultimate-carbon, oxal forming OH group: [SOS not sure if this translated correctly, a tricky one.] ![]() Under the action of a slightly energetic oxidation, the secondary alcohol group CHOH is converted into an acid group CO2H, the bicarbonated chain CH2 – CO2H becoming detached from the molecule, which decreases by 2 the number of atoms of carbon from it.
Under the action of a slightly energetic oxidation, the secondary alcohol group CHOH is converted into an acid group CO2H, the bicarbonated chain CH2 – CO2H becoming detached from the molecule, which decreases by 2 the number of atoms of carbon from it. ![]() Depending on fermentation conditions, the chain would be cut into more or less short sections. Finally, raw materials used in the composition of musts provide certain higher fatty acids (palmitic acid, oleic acid, etc.), which, being volatile with water vapor, can be found in the distillate.
Depending on fermentation conditions, the chain would be cut into more or less short sections. Finally, raw materials used in the composition of musts provide certain higher fatty acids (palmitic acid, oleic acid, etc.), which, being volatile with water vapor, can be found in the distillate.
Esters. — If, during distillation and storage of alcoholic liquids, a certain quantity of esters is produced chemically, it is especially during fermentation that these compounds are produced by biological means.
Kayser and Demolon (1909) observed that the quantity of volatile esters formed during fermentation was substantially proportional to that of the volatile acids produced by yeast; that the addition of acetic acid, either before fermentation or immediately afterwards, in no way increased the proportion of the esters; and, finally, that, by distilling the yeast collected immediately after fermentation, a certain quantity of esters was always obtained, it was concluded that esters were endogenous formation products.
This opinion, already issued by Ashby in 1907, was confirmed by the work of Ribereau-Gayon and Peynaud, Chai-Heung-Kim (2), and so on. It is currently accepted that esterification, physiologically during fermentation is carried out under the action of a special diastase, esterase.
(2) Enzymologia, VI, 3, 183, 1939.
Esters formed gradually diffuse into the liquids, especially at the end of fermentation, and can be destroyed over time by the yeast itself. Highly volatile products such as ethyl acetate disappear for a very high proportion, either by entrainment of carbon dioxide, or especially by evaporation in contact with air (3) (Kayser and Demolon).
(3) The vapor pressure of ethyl acetate at ordinary temperature is about twice that of alcohol, although the boiling points of these bodies are very close.
The nature and proportions of esters produced depend chiefly, as Kayser has shown, on yeast and bacteria races. Nitrogen nutrition of yeast and acidity are also important.
The dominant ester is ethyl acetate, which often forms up to 98% of the total esters. Ethyl formate can thus exist in significant amounts, especially in molasses musts, as well as ethyl butyrate. Finally, there are small amounts of high molecular weight fatty acid esters (caproate, caprate, laurate, etc.).
Ashby isolated from Jamaican molasses, a very slow fermenting yeast, giving per liter of alcohol at 100° to 18,000 mgr esters, consisting mainly of ethyl acetate. N. Deerr found in a Natal molasses a race of Monilia providing, in pure culture, by liter of alcohol 7,550 mgr of esters, mainly formed by acetate or ethyl butyrate.
[SOS not sure of this last line. Does he mean ethyl acetate AND ethyl butyrate?]
Kayser has also observed that some yeasts, are likely to provide very large quantities of esters. This author obtained the following results, by fermenting molasses must at 14% (by volume) with, on the one hand, a low yeast [bottom fermenting?] of Guadeloupe (A), and on the other hand the same yeast associated with a film yeast from Réunion. The analysis was done after 15 days.
[Percival Greg’s yeast #18 was a top fermenting Pombe yeast that formed a yellow head. It could be what they are describing. These guys were eye witnesses to a lot more variety.]  In another trial, by seeding the same molasses must with a combination of low yeast and film yeast simultaneously (A); with the low yeast and, once fermentation is in full activity, with the high yeast (B), the results were as follows (analysis made after 3 weeks):
In another trial, by seeding the same molasses must with a combination of low yeast and film yeast simultaneously (A); with the low yeast and, once fermentation is in full activity, with the high yeast (B), the results were as follows (analysis made after 3 weeks):
[I think the distinction here again is top fermenting and bottom fermenting.]  The following experiment by the same author shows the influence of nitrogen supply on production of volatile acids and esters. Molasses musts, which contained per liter: 150 gr of sucrose, 1 gr of acid phosphate of K and traces of Mg sulphate, was added 0.5 gr of nitrogen in the form of eau de touraillon [this may have been water from grains used as a nutrient], peptone, asparagine, leucine, urea, glycine and sulphate of ammonia. Yeast 3 was a film yeast isolated from a Reunion cane molasses, and yeast 5 another film yeast isolated from pineapple. The liquid was analyzed after 15 days of fermentation.
The following experiment by the same author shows the influence of nitrogen supply on production of volatile acids and esters. Molasses musts, which contained per liter: 150 gr of sucrose, 1 gr of acid phosphate of K and traces of Mg sulphate, was added 0.5 gr of nitrogen in the form of eau de touraillon [this may have been water from grains used as a nutrient], peptone, asparagine, leucine, urea, glycine and sulphate of ammonia. Yeast 3 was a film yeast isolated from a Reunion cane molasses, and yeast 5 another film yeast isolated from pineapple. The liquid was analyzed after 15 days of fermentation.  These results show that yeast nitrogen supply has a very clear, but also very variable, influence on quantities of volatile acids and esters obtained. The proportions existing between different acids and esters are also influenced (Kayser).
These results show that yeast nitrogen supply has a very clear, but also very variable, influence on quantities of volatile acids and esters obtained. The proportions existing between different acids and esters are also influenced (Kayser).
Lindner, studying 12 pressed yeasts, found that esters were formed mostly in well-aerated liquids, low in nitrogen, or when yeast growth was stimulated by a large excess of sugar or when fermentation took place at high temperature. The property of producing aromatic esters was manifested mainly in yeasts of secondary fermentation, living at the expense of old globules. [old globules may be old yeast cells?]
Finally, it should be pointed out that hydrogen sulphide resulting from yeast reduction of sulphites or sulphates in the culture medium can, under certain conditions, give ethylsulfhydric or mercaptan ester with alcohol. Presence of the latter has been reported in some wines when fermentation took place in the presence of sulfur, sulphurous acid, or sulphate. Production of mercaptan depends on yeast race and conditions of the medium: it increases with temperature and presence of easily reducible products. It appears to take place especially after the active phase of fermentation, when yeast have a more energetic esterifying action.
Transformation of sugar according to the theoretical formula of Gay-Lussac would give, for various fermentable carbohydrates, the following yields in alcohol (liters of pure alcohol per 100 kgs of sugar):  However, as it always happens, beside transformation of sugar into alcohol, the “fermentation derivative”, the importance of which varies with operating conditions, the actual yield is always lower than the preceding figures.
However, as it always happens, beside transformation of sugar into alcohol, the “fermentation derivative”, the importance of which varies with operating conditions, the actual yield is always lower than the preceding figures.
Pasteur (1), after several careful evaluations, had come to the conclusion that about 6% of sugar was used for purposes other than alcohol production. Operating with 100 gr of sucrose, dissolved in an appropriate mineral medium, he found as duplicating products (in gr.):
(1) Pasteur (L.) – Mémoire sur la fermentation, alcoolique. Ann. Chim. Phys. LVII et LVII. 1859.  Surplus of 5.65 gr of the 100 gr sugar introduced comes from water fixation that accompanies sucrose hydrolysis. 100 gr of sucrose thus giving 105.21 gr of glucose, there remains a small difference of 0.39 gr, which may be due to the hydrolysis of some by-products during fermentation.
Surplus of 5.65 gr of the 100 gr sugar introduced comes from water fixation that accompanies sucrose hydrolysis. 100 gr of sucrose thus giving 105.21 gr of glucose, there remains a small difference of 0.39 gr, which may be due to the hydrolysis of some by-products during fermentation.
Although representing an experimental result, the Pasteur figure (64.33 liters of alcohol per 100 kgs of sucrose) has been considered by some authors as a “theoretical yield”, impossible to exceed and even to achieve under the conditions of industrial practice.
It has been found, however, that the rate of by-products of fermentation can be reduced under certain conditions. Effront (1) in particular showed that the use of yeasts accustomed to hydrofluoric acid made it possible to reduce the amount of glycerine produced to one tenth of that obtained using ordinary yeasts.
(1) Influence de l’acide fluorhydrique et des fluorures sur les levures de bière. C. R. CXVII, 183 — CXVIII, 1894.
On the other hand, an appreciable part of the sugar diverted from alcoholic production is used for construction of yeast cells (cellulose and indeterminate matters of the Pasteur balance). Duclaux (2), on the basis of Pasteur’s work, estimates the weight of sugar required for formation of 1 gr to be between 1.5 and 2 gr dry yeast. According to Claassen, this figure would be equal to 2 gr. However, it is possible to save the sugar used for cell reproduction. Brown and Pedersen have indeed shown that the amount of yeast used to inoculate a must has no influence on the final number of cells, which remains about the same per unit volume. This maximum number of yeasts has been called “Brown’s Limit Number”, or “Specific Cell Saturation” (Boinot). If a quantity of yeast seed containing a number of cells close to the limit number is introduced into a must, little or no new cells are produced, and construction costs are consequently very low or nil.
(2) Traité de Microbiologie : S t. III. La fermentation alcoolique, Paris 1905.
It was objected that the total expenditure on maintenance of yeast cells was therefore greatly increased (Bettinger). The number of cells per liter of must is usually, in fact, in the usual methods of fermentation, 150 billion, which corresponds to 3.1 gr dry yeast. Under certain conditions, especially when using antiseptics, the figure can be lowered to 100 and even 80 billion (2 gr and 1.67 gr of dry yeast). Now, if fermentation is made by seeding the must with a quantity of cells corresponding to Brown’s limit, the number of cells is increased to 550 billion (11.46 grammes of dry yeast per liter), according to the observations of Grimaud.
This objection has been answered that the additional amount of sugar used in this case for unproductive maintenance expenditure is, however, much less important than that used in other processes for cell multiplication (3). In any case, Boinot (4), by fermenting in the laboratory various samples of beetroot molasses from France and cane molasses from Cuba and South Africa, by recovery of yeasts, was able to obtain yields varying between 64.5 liters and 66.9 liters of alcohol per 100 kgs of metabolized sugars (expressed as sucrose), all exceeding the Pasteur coefficient.
(3) By the method of yeast recovery, fermentation duration is, in fact, reduced by about 40%. In addition, it appears that the unitary maintenance expenditure is even higher as the number of cells decreases (Pérard).
(4) Bull. Ass. Chim. LVI, 728, 1939.
To the above causes of loss are added, in industrial fermentations, those due to intervention of foreign ferments (wild yeasts, mycoderms, bacteria), which attack the sugar but give little or no alcohol: conditions (temperature, acidity, composition of musts, etc.) adversely influencing the alcoholic function of yeast; the presence in the must of non-fermentable sugars up to 3-4% of total sugars); to evaporation and entrainment by carbon dioxide (1% or more of alcohol produced), when fermentations are made in open tanks. Also, yields generally remain very much lower than the Pasteur coefficient. In the case of cane molasses processed by various fermentation processes, the following percentages of the Pasteur coefficient would be obtained according to Magne:  In Jamaica, in the manufacture of full-bodied rums, yield is only 55% of the Pasteur figure. Some losses, due to the formation of acids and aromatic esters, are also unavoidable. Floro, following observations he has made, divides the various losses as follows:
In Jamaica, in the manufacture of full-bodied rums, yield is only 55% of the Pasteur figure. Some losses, due to the formation of acids and aromatic esters, are also unavoidable. Floro, following observations he has made, divides the various losses as follows:
[I think Floro is M.B. Floro who wrote important articles about Jamaica rum in response to Arroyo regarding distillery losses for full flavored rum production.]
Losses that can be reduced
Alcohol in the residual sugars of the must . . . . . . . . . . . . . . 7.65%
lost through evaporation and entrainment . . . . . . . . . . . . . . 6.44
in the bottoms and overflows . . . . . . . . . . . . . . . . . . . . . . . . . 1.78
in the vinasses . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 0.041
in leaks, etc. . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . 0.293
Losses due to your quality
Losses due to your quality . . . . . . . . . . . . . . . . . . . . . . . . . . . . 17.20%
lost after the end of fermentation and before distillation. . 9.33
esterified during distillation . . . . . . . . . . . . . . . . . . . . . . . . . . . 1.80
Total . . . . . . . . . . . 44.543
In Martinique, yield in the winery usually varies, in the case of molasses rhummeries, between 65 and 80% of the Pasteur figure. It is higher in vesou distilleries, where it reaches 75 to 90%, and even exceptionally 93%. Distillation losses are also quite high. The proportion of alcohol in the vinasse usually varies between 0.5 and 2 p. 1000, i.e. 1 to 4% of the quantity produced (in well-developed equipment, however, the alcohol content of the vinasse may drop below 0.2%). It is necessary to add the losses in the bottoms of the tank, which are thrown in the river. It is admitted that the average yield is, in vesou distilleries, from 100 to 55 per ton of cane handled.
Under very favorable conditions, however, it would be possible to go beyond Pasteur’s performance in industrial practice. Thus Russy Grimaud’s beet distillery (1), using the process of yeast recovery, obtained per 100 Kg. of sucrose implemented 63.83 liters pure alcohol, a figure increased by the alcohol lost to evaporation and entrainment by carbon dioxide (1 to 2%) and that it would have been possible to recover by fermentation in closed vats or by washing the gases. Pérard (2), at the distillery of Baleycourt, operating in open vats by the same process of yeast recovery, observed the average yield, for the 1936-37 season, of 63.97 liters alcohol per 100 kg of sucrose used. In the cane molasses distillery at Puerto Rico, application of the Arroyo process with thick musts would have been possible with certain vats to reach Pasteur’s yield (see Chapter V).
(1) Various processes for recovering alcohol lost in the vat room: Yeast recovery process. C. R 8. Cons. Int. des Ind. Agr., 1935. (2) Bull. Ass. Chim. LV, 212, 1938.
Acetic fermentation.
Kutying, in 1837, for the first time attributed production of acetic acid to the action of a microorganism. This theory, fought by Liebig and Berzelius, was definitively established by Pasteur in 1860. In 1906, Buchner isolated acetic bacteria diastase determining oxidation of alcohol, oxidase.
Mechanism of fermentation.
The reaction which gives rise to acetic acid was first considered to correspond to a simple oxidation, according to the formula:
CH3-CH2OH+02 ―> CH-CO2H+H20
The phenomenon is actually more complex. It is currently accepted that the first stage of the oxidation of ethyl alcohol would be acetaldehyde, which was isolated by Neuberg and North as a combination of sulfite:
CH2 – CH2OH +O2―> CH3-COH + H2O [Need to recheck these atom counts]
According to Neuberg and Windisch, a sort of reaction from Cannizaro would occur, giving rise to acetic acid and alcohol, which would enter the cycle to be oxidized again:
2CH3 – COH+H20 ―> CH3 – CO2H+CH3 – CH3OH
Kluyver and Donker, inspired by the theory of Wieland (1), considering acetic fermentation as an oxidation-reduction phenomenon.
The first step would be dehydrogenation of alcohol, giving rise to acetaldehyde, which after hydration would in turn undergo dehydrogenation:

Oxygen, necessary for the action of the acetic ferment, acts only as a hydrogen receptor:
2H2 + O2 ―> 2H2O
Conversion of ethyl alcohol to acetic acid could be achieved anaerobically by replacing acetic bacteria with a hydrogen receptor (platinum black, palladium, methylene blue).
The theory of Kluyver and Donker helps explain the rapid progress of fermentation. The reaction indicated by Neuberg and Windisch, however, remains possible, but would only be a side reaction.
In addition to aldehyde and acetic acid, secondary products are still poorly known during acetic fermentation, such as fixed (lactic, succinic) or volatile acids (caproic, valerianic, etc.), and esters (ethyl acetate, etc.). On the other hand, some bacteria completely oxidize alcohol, giving carbon dioxide and water.
[Secondary products are where there may be a great benefit to rum. Note that lactic acid is considered a fixed as opposed to volatile acid.]
Acetic ferments can also attack alcohol other than ethyl alcohol, as well as sugars. Oxidizing power varies greatly depending on the species. Propyl and butyl alcohols are converted into propionic and butyric acids, glycerin into glycerol acid and dioxygenone. In sugars, the aldehyde function is transformed into an acid function: for example, glucose gives gluconic acid and, with certain bacteria, oxalic acid. Sucrose, levulose, and generally ketones are not attacked (Bertrand and Watermann).
[This paragraph represents a very large hole in modern understanding. Can this ever be used to process fusel oil?]
Acetic ferments.
Acetic bacteria are formed by net-shaped cells, often joined in chains and generally immobile. They usually develop on the surface of alcoholic liquids in obligatory aerobiosis, forming at first thin veils, then becoming more or less thick, transparent or opaque, oily or dry, more or less wrinkled. In the young state, the veil is difficult to wet; once old, it breaks easily and can immerse itself in the liquid, forming a mucilaginous mass (mother of vinegar). They do not form endospores and do not liquefy gelatin.
There are many species of acetic bacteria, differentiated by the size and shape of cells, the nature of the veil formed, the appearance of colonies on solid media, physiological properties (acid and alcohol resistance, optimum temperature, oxidizing power, etc…).
The species found in distillery musts are generally weak producers of acids, they do not tolerate alcohol well, but are able to oxidize many sugars. They most often give thick veils.
Sugar cane juices contain several species of acetic bacteria. Tanaka, at Formosa, was able to isolate the following, each represented by several varieties: Acetobacter (Bacterium) xylinum Brown, A. acetosum Henneberg, A. Lindneri Henneberg; Bacterium aceti Brown, B. curvum Henneerg; Gluconoacetobacter liquefaciens, G. Asai.
Ashby has encountered two distinct species in Jamaican distilleries: a) A bacterium that appears quickly in fermented musts with low or high acidity. It forms a delicate blue veil, which becomes white afterwards, but always remains fragile. In a glass container, the veil rises along the walls, above the surface of the liquid. The bacterium comes under the aspect of short, thick sticks, united in chains of short length and appearing yellow or yellowish brown when dyed by iodine. It resembles Bacterium Kutzingianum Hansen, except that it does not stain blue with iodine. b) A bacterium which produces a veil on the surface of liquids of cartilaginous consistency, very resistant. It is in the form of long, narrow sticks, staining blue with iodine and sulfuric acid, and can be identified with Bacterium xylinum Brown.
The first of these ferments multiplies vigorously when the alcohol content is between 7 and 9%. It gives about 4% of acetic acid, after a fortnight (7.5% maximum). B. xylinum, on the contrary, can not develop when the alcoholic richness reaches 7°; it produces about 3% acetic acid in liquids containing 4.5% alcohol. It is therefore found mainly in fermented cane juice, while the previous species is found in molasses musts containing 6% alcohol or more.
According to Watts and Tempany, acidification which affects freshly expressed cane juice, before alcoholic fermentation has developed (surissement du jus), is due to action of an anaerobic ferment, directly attacking sugar. Acidity formed accounts for about 2/3 of the fixed acids and 1/3 of the volatile acids, of which acetic acid predominates.
Importance in the distillery.
Acetification is a frequent condition in distiller’s musts. In the old days, when the conditions of its development were poorly known, it caused considerable losses, lowering alcohol yield and spoiling the quality of the product obtained (excessive content of spirits in acetic acid).
The main factors that promote acetic fermentation are: a) high fermentation temperatures, which reduce the vigor of alcoholic yeasts and are instead favorable to acetic bacteria. Optimum temperatures of propagation generally vary between 30° (Bacterium xylinum) and 36° (B. aceti); b) insufficient initial acidity (especially in the case of vesou musts), which is detrimental to the activity of the yeast; e) the low alcohol content of fermented musts; d) the use of contaminated yeast or the lack of cleanliness of the vat room.
Acetic ferments, when they do not exert an action too predominant, can intervene usefully in bouquet production of the spirits, especially those with developed aroma. Some practices used in rhummeries are intended to promote their development. This is the case of the preliminary treatment of defecation scum and cane juice in the presence of bagasse, in the manufacture of grand arôme rums in Jamaica. It should also be noted that in the past, producers of agricultural rum in Martinique sometimes abandoned the cane for several days after cutting before handling it, in order to obtain a more robust eau-de-vie.
Butyric and acetonobutyl fermentation.
The first observations on butyric fermentation were made by Pasteur (1861). The production of butyl alcohol by fermentation, reported by Fitz in 1876, was studied by Grimbert (1893) and Beijerinck. Duclaux showed that butyric and butyl fermentations were caused by the same organisms: when the artificially neutral medium is maintained by the addition of chalk, butyric acid is produced, whereas if the culture is allowed to acidify freely, normal butyl alcohol is formed. Finally, in 1912, Fernbach and Schoen found that besides butyl alcohol, acetone appeared in a definite proportion. [this may offer a clue to the usefulness of buffers in rum ferments.]
Mechanism of fermentation.
The mechanism of butyric fermentation is very complex and still poorly understood.
It is generally accepted that degradation of the sugar molecule follows the same course as in alcoholic fermentation, up to the acetaldehyde stage. Then, hydrogen, instead of being fixed to the aldehyde formed, stabilizes in the molecular state and emerges from the medium at the same time as carbon dioxide. Acetaldehyde undergoes a condensation which transforms it into aldol, or β-oxybutyric aldehyde: ![]()
In a neutral medium, hydrated aldol undergoes an intramolecular oxidation-reduction, which results in butyric acid:
![]() In acidic medium, according to Schoen, there would be, oxidation-reduction of 2 molecules of aldol to give alcohol β-oxybutyl and β-oxybutyric acid, by the reaction of Cannizaro.
In acidic medium, according to Schoen, there would be, oxidation-reduction of 2 molecules of aldol to give alcohol β-oxybutyl and β-oxybutyric acid, by the reaction of Cannizaro.
Oxybutyric acid would be oxidatively converted to acetoacetic acid, and oxybutyl alcohol, by concomitant reduction, to butyl alcohol. Finally, by decarboxylation, acetoacetic acid gives acetone and carbon dioxide:
 The general formulas of the reactions would be the following: a) Butyric fermentation:
The general formulas of the reactions would be the following: a) Butyric fermentation:
![]() (b) acetonobutyl fermentation:
(b) acetonobutyl fermentation:
![]() In fact, these formulas are not exclusive. In addition to the main phenomenon, there are deviations from fermentation, which give rise to various secondary products: ethyl and isopropyl alcohols, acetic, formic and propionic acids, etc.. The substances formed vary in nature and quantity according to the ferment considered, fermentable material, reaction of the medium, stage of fermentation (at the beginning of the acetonobutyl fermentation, a little butyric acid always occurs).
In fact, these formulas are not exclusive. In addition to the main phenomenon, there are deviations from fermentation, which give rise to various secondary products: ethyl and isopropyl alcohols, acetic, formic and propionic acids, etc.. The substances formed vary in nature and quantity according to the ferment considered, fermentable material, reaction of the medium, stage of fermentation (at the beginning of the acetonobutyl fermentation, a little butyric acid always occurs).
By way of example, the following composition of the products obtained by Buchner and Mesenheimer (1) can be obtained by fermenting, by Bacillus butylicus Fitz, 100 grams of glucose in the presence of chalk and salts:
(1) Ber, Deut. Chem. Ges. XLI 1410, 1910.
 Butyric ferments.
Butyric ferments.
Butyric ferments take the form of large batons, often polymorphous (one finds forms in club, spindle, etc.), and are movable. They sporulate easily keeping for a long time easily as spores (even at temperatures of 100-105°). These ferments are generally anaerobic, but in addition to obligatory anaerobes, facultative anaerobes (Aero-bacillus genus) have been found, occurring optionally in response to circumstance. They prefer neutral or alkaline media and high temperatures (35°-40°).
True butyric ferments use ammonia as a nitrogen source. This differentiates them from putrefying bacteria, which attack proteic materials. There are, however, between these two groups transitional forms.
Butyric bacteria can attack most carbohydrates, including starch. Cellulose, however, is not modified except by quite special species. Sugars are degraded either by oxidation or much more frequently by fermentation.
Butyric ferments are widespread in nature. They are encountered in milk, manure, soil, beet and cane juice, candy bars, and so on. There are many species, differentiated by their action on various sugars and proteic materials, by nature, and the amount of by-products they form, etc. Among those best studied are the following species:
—Bacillus butyricus Pasteur.— It ferments lactate of lime, the milk having previously undergone a lactic ferment, of glycerine, of certain sugars, giving rise, as principal products, to butyric acid, hydrogen and carbon dioxide in varying proportions. [I believe he is talking about lime water used in nixtamalization for making tortilla chips and stuff like that.]
—Clostridium butyricum Prazmowski.— Anaerobic and giving mostly butyric acid; it is the species that is most commonly found in distillery musts.
—Bacillus butylicus Fitz.— It ferments sucrose, glucose, mannite and glycerin, giving hydrogen, carbohydrates, lactic acid, succinic, butyric acid, butyl alcohol and a little ethyl alcohol.
—Bacillus orthobutylicus Grimbert, who ferment sugars, inulin, dextrin and starch, giving hydrogen, carbon dioxide, butyric and acetic acids and normal butyl alcohol.
—Bacillus tetryl Arroyo.— Discovered by Arroyo on the roots of the Kassaer cane in Puerto Rico. It has been used industrially for the manufacture of acetone and butanol from cane molasses. [This likely relates to Arroyo’s patented work before joining the experiment station.]
—Clostridium saccharolyticum Bergery, whose presence has been reported by Hall, James and Nelson in Barbados cane syrup.
—Clostridium saccharo–butyricum Arroyo.— Found in Puerto Rico on annatto seeds and used for the production of butyric acid from cane molasses. It gives, according to Arroyo, in addition to hydrogen and carbon dioxide, about 93% of normal butyric acid, 4.1% of acetic acid, 1.9% of propionic acid and 1% of higher fatty acids (caproic acid, heptoic acid, etc.). It does not produce appreciable quantities of alcohols, aldehydes or ketones. Activity of the bacterium is stopped when the concentration of sugars in the medium exceeds 6 gr per 100 cc. or when the alcohol level reaches 8% by volume. It is the same when the pH goes down to around 4.0.
Importance in the distillery.
Distillery raw materials normally contain butyric ferments, in the state of bacteria or, more often, spores (molasses). Activity of these microorganisms is annihilated, when the fermentation is carried out with pure yeasts or in the presence of antiseptics. On the other hand, they can play an important role in spontaneous fermentations, especially when they have a long duration. Their intervention results in a decrease in alcohol yield, and also in the production of higher alcohols (butyl, propyl, etc.) and volatile acids (butyric, propionic, formic, etc.), which contribute to bouquet formation in the eaux-de-vie.
Allan was able to isolate in Jamaica’s rhummeries, and more particularly liquids from the “muck hole”, various butyric bacteria. He found that if they do not get along well with cane juice alone, they develop vigorously in the addition of sugar solutions made of albuminoidal substances, as well as in vinasse with the addition of yeast extract. This last medium corresponds substantially, from the point of view of composition, to the must obtained by mixing vesou or molasses with vinasse. The bacteria grow well from 26° C, but have the optimum temperature 35°. The quantities of acids formed however remain rather low (0.3 – 0.4%), if the must is not neutralized, by means of lime for example. [always be buffering]
According to the same author, the bacteria predominate in the tanks towards the end of long-term fermentation in distilleries producing grand arôme rum, to the point where the yeasts of the fermented liquid disappear completely. Intervention of butyric bacteria explains that the fusel-oil of Jamaican rum is formed essentially by normal butyl alcohol.
Finally, Arroyo, recently in Puerto Rico, by fermenting musts of cane molasses with Pombé yeast in symbiosis with Clostridium saccharobutyricum, was able to obtain a rum having all the characteristics of Jamaican rums, as regards bouquet and chemical composition. This author has observed that by cultivating the bacterium in symbiosis with yeast, its multiplication and formation of alcohol were considerably accelerated. The duration of the fermentation of the yeast, from 70 to 96 hours in the case of yeast alone, was reduced to 28-48 hours. The author attributes this fact to the action of radiation emitted by the bacterium and comparable to the mitogenic rays of Gurwitsch (1).
(1) Mitogenetic radiation, discovered by Gurwitsch (Arch, Mikrosk Anat. und Entro Mech. C. 11, 1923), are issued by certain living organisms at certain stages of development. They pass through quartz, but not glass, and when they meet with other growing tissues, they can act upon them, accelerating their development or reproduction. Arroyo has found that various bacteria can emit similar rays, acting on the speed of multiplication and zymogenic power of the yeasts, even when the bacterium is separated from the yeast by a wall of quartz.
Lactic fermentation.
It was Pasteur who isolated, in 1857, the first lactic ferments. Today we can distinguish between true lactic ferments, which produce lactic acid almost alone, and lactic pseudo-ferments, which at the same time give rise to significant quantities of various by-products (acetic acid, ethyl alcohol, CO2, H, etc.).
Mechanism of fermentation.
It was once thought that production of lactic acid from hexoses was by simple splitting, according to the formula: C6H12O6 —> 2C3H6O3
But, during lactic fermentation, several of the bodies isolated in alcoholic fermentation (hexose-phosphate, acetaldehyde, methylglyoxat) have been found, the mechanism of these two fermentations is now considered to be analogous.
According to Neuberg, sugar would give rise, through hexose phosphates, methylglyoxal hydrate. This would then be transformed into lactic acid, by intramolecular oxidation-reduction (caused by glyoxalase):
 According to Meyerhoff, lactic fermentation could be compared to the production of lactic acid in the muscle, under action of lactic zymase (isolated from muscle in 1927). The evolution of sugars would be the same as in alcoholic fermentation, up to the pyruvic acid stage. It would then undergo an oxidation-reduction, in the presence of phosphoglycerol, to give lactic acid and phosphoglyceral:
According to Meyerhoff, lactic fermentation could be compared to the production of lactic acid in the muscle, under action of lactic zymase (isolated from muscle in 1927). The evolution of sugars would be the same as in alcoholic fermentation, up to the pyruvic acid stage. It would then undergo an oxidation-reduction, in the presence of phosphoglycerol, to give lactic acid and phosphoglyceral:
 Phosphoglyceral thus formed would react on a second molecule of pyruvic acid, to give still lactic acid and phosphoric acid, which would enter the reaction to regenerate the pyruvic acid.
Phosphoglyceral thus formed would react on a second molecule of pyruvic acid, to give still lactic acid and phosphoric acid, which would enter the reaction to regenerate the pyruvic acid.
This mechanism is that of true lactic fermentation. The transformations are much more complex and poorly known, in the case of pseudo-lactic ferments, which produce at the expense of sugars large quantities of gas (CO2 and H) and give rise to various other acids (acetic, succinic, formic) and ethyl alcohol.
Lactic ferments.
These ferments, very numerous, are in the form of batons of variable size or cocci, isolated, united two by two diplobacilli, diplococci), or arranged in more or less long chains (streptobacilli, streptococci); they are immobile and do not form spores.
Some species are anaerobic, others are aerobic or anaerobic, others are indifferent to oxygen. They are sensitive to acidity: they produce only 2% maximum lactic acid and generally much less. They prefer neutral environments. Not sporulating, they are usually destroyed by heating at 65-70° for 5 minutes. Optimum growth temperature may be relatively high for some species (40-50° for bacteria of the genus Thermobacterium), lower for others (30°). True lactic ferments require peptones for their nitrogen nutrition; the lactic pseudo-ferments are content with amino acids or with ammoniacal salts. Very common in nature, they are found in milk, distiller’s musts, manure, etc.
Lactic ferments found in the distillery belong especially to the group of lactobacilli, anaerobic rod-shaped bacteria, often joined by 2 or in chains, able to withstand high doses of lactic acidity and giving only traces of products other than lactic acid.
Lactabocillus Delbruckii Leichman and L. Lindneri were isolated from cane molasses.
The first of these organisms is in the form of rods of 2.7 mus to 8 mus long and 0.4-0.7 mu wide, either isolated, or combined 2 by 2 or sometimes very long chains. It does not grow in milk and thrives especially well in non-hopped wort and distillery musts. Optimum temperature is 45°. The amount of acid formed is up to 1.6%. Acidification is slowed by 4% and stopped by 10% alcohol.
Lactic acid bacteria meet quite frequently in rhummerie musts (Ficker and Szügs). They would predominate in the fermentation of defecation scums (Ashby). A representative of the group of pseudo-lactic ferments, Leuconostoc mesenteroides, is easily developed in vesou or molasses musts with neutral or alkaline reaction.
Lactic acid bacteria must generally be considered as a fermentation disease, the development of which is favored by high fermentation temperatures, insufficient acidification of musts, lack of cleanliness. However, secondary products (acetic, formic, etc.) to which various species give rise can play a useful role in aroma production.
In the distillery of starchy material, lactic acid bacteria are used to obtain must acidification. These are left to spontaneous lactic fermentation, at high temperatures (50-55°), or sometimes seeded with a pure culture of Lactobacillus Delbruckii.
[Sour mash whiskey process]
Mycodermic fermentation
 In this fermentation, which occurs easily if the vats are left to their own after the end of the alcoholic fermentation, alcohol is completely oxidized, with production of water and carbon dioxide.
In this fermentation, which occurs easily if the vats are left to their own after the end of the alcoholic fermentation, alcohol is completely oxidized, with production of water and carbon dioxide.
The agents of this fermentation are asporogenous yeasts, of the genus Mycoderma. These are most often in the form of elongate cells, cylindrical, transparent protoplasm and vacuolized, showing a tendency to remain united in thin chains. They are aerobic and form on the surface of liquids, from the beginning of the fermentation, a folded veil, filled with air bubbles (levures à voile).
Mycoderms are very common in the air and live mostly in liquids containing alcohol. Many species have been described. Of particular note is Mycoderma cerevsiae Desm, which is found in breweries and alters beer as well as Mycoderma vini Desm, which causes the flower of wine.
While some species do not tolerate more than 1%, others support 15% alcohol. Mycoderms prefer mediums rich in organic matter. They poorly support acidity, their growth usually being stopped by 2% acetic acid. However, they can attack organic acids (acetic acid, malic acid, tartaric acid, etc.) if the concentration of these acids is low. Rarely, do they ferment some sugars, giving a little alcohol.
The main products of fermentation are water and carbon dioxide. Secondary products include small amounts of organic acids (formic, succinic, maline, acetic, etc.) and esters (ethyl acetate).
Mycoderms, if they play a harmful part in burning alcohol formed, may in certain cases intervene happily, producing aromatic principles. It is to these ferments that the flavor of certain sherry wines is due, which age several years in barrels unfilled and covered with Mycoderma vini. Note also that in the rhummerie, to obtain a more “bouquetéd” eau-de-vie, vats are sometimes allowed to “blanch”, that is to say, to cover themselves with a mycodermic veil, before proceeding to their distillation. In the manufacture of certain grand arôme rums, it is the rule to abandon the vats for 3 or 4 days after the end of the alcoholic fermentation.
Putrid fermentation.
Putrescent ferments are distinguished from the ferments previously studied by the fact that they cause an alkalinization of the medium: nitrogenous matters are decomposed first into amino acids, then into various volatile acids (carbonic, sulphohydric, acetic, propionic, butyric, valeric, etc.), which combine with the produced ammonia. There is also release of aromatic or heterocyclic products. Aerobic bacteria push further degradation than anaerobic species. The nature of the attacked amino acid, the conditions of the fermentation intervene, at the same time as the microbial species, to modify the nature of the products formed.
Many putrid ferments also attack sugars and some of them, which are transitional species with lactic and butyric ferments, give rise to free fatty acids. The action of these organizations on sugars is, however, very limited.
We usually divide fermentations of putrefaction in anaerobic bacteria, which develop in depth, and in aerobic bacteria that form veils on the surface of liquids. The former are close to butyric pseudo-fermentations, of which they are distinguished by their property of attacking albuminoid substances. The latter are mainly represented by the groups Bacillus fluorescens, B. proteus, and B. subtilis.
Fluorescent bacteria, which are very common in water, air and the upper layers of the soil, are mobile, non-sporulating, usually gram-negative, indol-producing.
Those of the proteus group are generally mobile, very polymorphic and not sporulating. They energetically attack proteic matters and ferment sugars, with production of hydrogen, carbon dioxide, acetic and succinic acids. The presence in ferments of this group has been reported in the manufacture of Jamaican rum, and in particular in the liquid of the “muck hole” (Allan).
Bacillus subtilis Ehrenberg is a mobile, rod, quite often polyphobic, gram-positive bacteria; it produces very heat-resistant spores and has an optimum development temperature of about 40°. The final molasses of Cuba, along with B. subtilis properly so called, have been found to contain Bacillus vulgatus Migula, B mensentericus Trevisan, B. mensentericus fuscus Flugge, B. liodermos Flugge, and B. levaniformans Greig Smith.
Putrid bacteria usually have a very minor importance in the distillery. Although they are commonly found in certain raw materials (molasses), their action on sugars and albuminoid substances remains very limited, because of their sensitivity to acids; it is rare to see putrefying bacteria withstand a pH below 4.
In some cases, however, they can intervene by destroying a certain amount of sugar and giving rise to aromatic products acting on the bouquet of the eau-de-vie, favorably (production of certain volatile fatty acids), or unfavorably (production of H2S, indol, etc.). It would play a role in the production of Jamaican grand arôme rum (Allan, Cousins).
Gummy fermentation.
In this fermentation, sugar juices are transformed into a gelatinous and viscous mass, under action of various microorganisms which produce, from sugars, gummy substances known under the names of dextran and levulan. These substances have been assigned the same crude formula as cellulose, (C6H10O5)n, but they correspond, according to some authors, to hydrated products of variable composition.
Dextran is a colorless gum that swells in water and dissolves in alkaline liquors. It is dextrorotatory (αⁿ=+200°) according to Scheibler and gives by hydrolysis of glucose.
Levulan has the appearance of a gum which becomes brittle by desiccation. It melts around 250°, dissolves in hot water and gives by cooling, a gelatinous mass. Its rotatory power is equal to 221°. It provides by hydrolysis of levulose. It does not reduce, no more than dextran, Fehling’s liquor.
Frai de grenouille. [frog eggs]
The most well-known microorganism producing gums is Leuconostoc mesenteroides Van Tieghem, which determines the affliction for a long time known as gomme de sucrerie, or frai de grenouille. This disease, reported as early as 1822 by Vauquelin in bottles of cane juice shipped from Martinique in France, appears both in beet sugar (mainly when sugar beets are not ripe enough) and in cane sugar (Java, Hawaii, Cuba, Louisiana, etc.). In Louisiana, canes damaged by frost often undergo, at the arrival of warm weather, gummy fermentation.
Affected sugar juices are transformed into compact gelatinous masses composed of insoluble, colorless or pink lumps, immobilized in a viscous liquor. These masses, of firm and elastic consistency, resemble frai de grenouille.
Leuconostoc mesenteroides is a streptococcus, in the form of spherical grains surrounded by a glairy sheath (dextran). In the young state, it affects the appearance of a string of small grains surrounded by gelatin, then long refractory puddings, which curl irregularly while keeping in their axis the rosaries of grains that gave birth to them. Different tubes, curling up on themselves, form mucous bodies, constantly enlarging, and whose contoured surface resembles that of the brain, then masses more and more voluminous. The fermentation terminated, the grains soften and the rosaries break up.
Leuconostoc, which has been found in water and soil by Berijerinck, prefers alkaline media; its development is slow in neutral or acidic juices. The optimum temperature is around 36°; however, it develops mainly at relatively low indoor temperatures at 20°, unfavorable to the action of yeasts (Owen).
Leuconostoc attacks sucrose and glucose, with production of dextran, mannite and fatty acids (lactic, etc.) with lactose and maltose, it gives rise to lactic acid, but without forming a sheath. It secretes sucrase, which inverts to sucrose.
According to Sacchetti, “gomme de sucrerie” is due to the symbiosis of two microbes, Leuconostoc mcgenteroides and Bacillus vulgatus Migula, giving birth first to dextran and the second to levulan, and a yeast, Saccharomyces cartilaginosus Lindner. This symbiosis was confirmed by Monoyer, who described the yeast associated under the name of Torulopsis Canbresieri Mon. n. sp. Leuconostoc is found in the central part of the gummy particles, the bacillus in the middle part, while the yeast forms an outer crust, which protects the bacteria against action of dissolved oxygen in the liquid. Various species of Coccaceae similar to Leuconostoc have been studied by Schöne, Zettnow, and Gonnerman. They are differentiated by their size, their behavior on solid or liquid media, nature of products formed, etc.
Other producers of gum.
Various other microorganisms that produce gums from sugar have been described. Let’s mention the following:
—Bacterium gelatinosum beta Glaser, which forms a mucous veil on the surface of beet juice, much like that produced by Leuconostoc. It liquefies gelatin and interchanges sucrose, Optimum temperature: 40°.
—Bacillus viscosus sacchari Kramer;
—Semiclostridium commune Maassen, which forms levulan at the expense of sucrose, but does not attack the invert sugar.
—Bacillus levaniformans Greig Smith. Found by Greig Smith (1) in 1901, in cane juice and raw sugars in Queensland, the microbe, like the previous one, attacks only sucrose, with levulan production. The same is true of Bacterium gummosum Ritsert.
(1) J. Chem. Ind. XXI, 1381, 1902.
Semiclostridium commune forms sheaths like Leuconostoc, but the others do not produce them.
Following Owen (2), the bacteria of the group mesentericus (bacilli of the potato): Bacillus mesentericus, B. vulgatus. B. liodermos, acquire the property, if grown for several generations in saccharide media, to transform sucrose into levulan. These microbes, which are normally found in soil, can acquire this property in the neighborhood of sugar houses. Introduced in the juice by bagasse and particles of earth adhering to the canes, they are found in press cakes and final molasses, because they are likely to resist, in the state of spores, the high temperatures of sugar manufacture. Press cakes are commonly used as fertilizers and the molasses bacteria also return to the soil when they are used for feeding animals, resulting in a gradual acclimatization of the enzymes to sugary solutions and a dissemination of these organisms in the soil of cane plantations.
(2) J. Bacteriology VIII, 421, 1923.
Owen considers that many gum-producing microorganisms, described by authors as distinct species, are actually only forms derived from bacteria of the mesentericus group. This would be the case with Bacillus levaniformans, which would become B. mesentericus levaniformans.
These ferments produce levulan directly from sucrose: the previous inversion of it by sucrase decreases and slows down the formation of gum. They prefer alkaline media, the optimum pH being between 6.7 and 7 (Owen).
Cellulose production.
Browne reported that in Louisiana, an aerobic fermentation giving rise to cellulan is quite common. Cane juice forms gelatinous masses, which can weigh several pounds and have a stratified appearance.
The membranes constituting the strata are of cellulosic nature. Under the microscope, they appear to be formed by chains of bacteria, intermingled with streptococci embedded in a cellulosic sheath. Bacteria are usually mixed with yeast cells. The cellulan producing organism is very similar, if not identical, to Bacterium xylinum.
Fermentation by the mucédinées [molds].
The presence in cane molasses of many moldy sugars has been reported. Church and Thom, for example, found the following species: Aspergillus niger Van Tieghem, A. flavus Link, A. nidulans Winter, Aspergillus sp. (blue); Citromyces spp., Penicillium expansum, P. diverticatum, P. luteum, P. roseum, P. purpurogenum.
Molds develop on the other hand as parasites on sugars, sometimes even on vats and walls of poorly maintained distilleries.
These organisms attack a variety of foods. They can notably metabolize sugars, giving carbon dioxide and water. Between this final term and the ternary compounds, we find various intermediate bodies, corresponding to incomplete oxidations: saccharic, oxalic, citric gluconic acids, etc. These acids usually appear only fleetingly and are soon metabolized in their turn. But under certain conditions, they can remain in the liquid and contribute to bouquet formation.
Among the most interesting products of this fermentation are oxalic acid, which appears mainly in nitrogen and sugar-rich media (15-20%), and citric acid, which is produced now industrially by molasses fermentation (Cahn process).
Some molds, mainly belonging to the family Mucoraceae, can saccharify starchy materials and even, under certain conditions, give alcohol. They are the basis of fermentation processes practiced in the Far East. In this part of the world they play the same role as malt in Europe. [koji]
Particularly noteworthy are the following species:
—Mucor (Amylomyces) Rouxii Wehmer, ferment of the annamite yeast (same), used in the preparation of annamite rice alcohol or Ru’o’on (vulgarly choum-choum). It is also found in the yeast used in China.
—Aspergillus (Eurotium) oryzae Cohn, ferment of Japanese yeast (koji), used in the fermentation of a rice beer (sake).
—Mucor Prainii Chodat et Nechitch, encountered in the yeasts of India (ranu, murcha).
—Mucor oryzae Went, Mucor javanicus Wehmer and Rhizopus oryzae Went and Prinsen-Geerligs, microorganisms of ragi, yeast used in the Dutch Indies, in the preparation of Batavia arak.
These molds, living aerobically, transform the starch of grains and rice into glucose. Some of them (M. Rouzii, M. javanicus, etc.), when grown in anaerobiosis, can also produce alcohol, but low amounts (5 to 7% alcohol maximum) and very slowly. Transformation of sugar into alcohol is especially the work of yeasts, which still exist in abundance in the yeast of the Far East.
The properties of Mucors in distillery of starchy materials have also been used in Europe, and more particularly in the manufacture of grain alcohol (Amylo process, developed by Collette and Boidin). Amylomyces Rourii was first used, followed by various others more active or resistant Mucors (Mucors B, Boulard, Delemar, etc.).
Nitrous fermentation.
Nitrous fermentation is quite common in juice and beet molasses distilleries. It results from the development of reducing bacteria, which decompose nitrates of the must, giving mainly nitrogen gas and nitrites. The latter are themselves decomposed by the acids with production of nitrogen dioxide, which is transformed in contact with air into vapors sparkling with nitrogen peroxide: the tank is covered with yellowish bubbles. Butyric ferments can also cause, thanks to the hydrogen they emit, reduction of nitrites to nitrates and the formation of nitrogen peroxide.
Nitrous fermentation has not been reported in Cannes distillery. Bassières (1) however noted in Martinique a case of “nitriform fermentation”, where the vats were covered with large bubbles of a yellow rust and a kind filth of the same color. He could not show the presence of nitrites.
(1) in Kervégant – L’industrie Rhumiere à la Martinique, Bull. Agr. Martinique 11, 29, 1933.
2 thoughts on “Kervegant Chapter III Distillery Fermentations”