Follow along: IG @birectifier
Kervegant Chapter II Raw Materials of the Rhummerie: [PDF]
Pages 28 – 66
[This chapter has fascinating writings on skimmings and explains where they disappeared to in the modern era.]
Chapter II
RAW MATERIALS USED IN THE RHUMMERIE
The raw materials used in making rum are, in order of importance: molasses, cane juice and syrups. Various other residual sugar products such as defecation scums, apparatus washing water, etc. are also possibly sent to the distillery. Finally, vinasses resulting from the distillation of fermented liquids frequently enter, in more or less important proportions, into must composition.
Cane juice
It is only in the French West Indies that cane juice, or Vesou, is used on a large scale in rum production. Elsewhere, it is used for this purpose only in an exceptional way and in order to obtain a product most often consumed locally.
In Martinique and Guadeloupe, vesou rum is produced in special establishments treating cane only for distillation, the “rhummeries agricole”. Exceptionally, the sugar factories send juice coming from spoiled or insufficiently ripe canes directly to the distillery, which, because of their high glucose coefficient, would give bad results in sugar. It has also sometimes been recommended to reserve for distillation, juices of the last mill, which are richer in non-sugar and ferment more easily than those of the first mills.
Extraction of the vesou.
The primitive extraction system of cane juice was the vertical mill with 3 rolls placed in line. The median cylinder received the movement of a carousel shaft in the air, either from oxen or mules, from a gear wheel, from a windmill, or from a steam engine, and transmitted it in the opposite direction to the other two side rolls by toothed wheels.
A worker presented cane rods between the middle cylinder and one of the side cylinders. Another worker, placed on the opposite side of the mill, received the pressed rods and immediately directed them between the middle roll and the other lateral roll, very close together, so that they stand out on the side of the first maneuver. The yield in vesou was only 50% of the weight of the canes used. The rate of ligneous of the old varieties of cane (Bourbon, Crystalline) being of 10% on average, there remained about 40% vesou in the bagasse.
The vertical mill has practically disappeared today; it is no longer in use except in some primitive installations of the Antillean or South American countryside.
It has been gradually replaced, since 1840, by the horizontal mill which has 3 rolls arranged in a triangle. In small distilleries, the mill is short, of low power and constituted by a single train of cylinders. In larger plants, it is formed by 2 or 3 sets of cylinders, joined by a bagasse transporter and sometimes preceded by a shredder. The most common brands in the French West Indies are mills from Mariolle, Gilain, Society of mechanical constructions of St-Quentin, Thiriau, Company of Fives-Lille, Fletcher. Feeding is generally ensured by means of a rod-lift, constituted by a system of two endless chains interconnected by wooden or metal slats.

FIG. 4. – Vertical windmill with carousel in the air (Martinique, 19th century).
Mills at agricultural distilleries are still quite often driven by hydraulic wheels. However, large distilleries are equipped with bagasse-heated steam engines, capable of moving powerful mills, which make it possible to obtain an extraction almost as good as in the sugar factories.
Imibition is a widespread practice. If the mill has only one train of rolls, the bagasse is subject to repressing, after being watered or macerated in water for 15 & 20 minutes. When the mill is double, imbibition water is distributed, usually by means of a perforated ramp, at the outlet of the first mill. Finally, when it is triple, bagasse is watered after the first mill with the small juice of the 3rd pressing and, at the exit of the second mill, with water. When this is lacking, it is sometimes used for the hot or cold imbibition of vinasse.
The imbibition rate is much higher than in the sugar industry. Frequently, the amount of water is adjusted so that the mixed juice has the density required for the final “composition” of the must, i.e. about 1.040 S.G.. It varies consequently during the rhum season, following the saccharine richness of the cane.
In some distilleries, imbibition is replaced by maceration: bagasse instead of being watered, goes into a bath of water or small juice, or it lasts 10 to 20 minutes before being re-pressed. This system, which has been abandoned in cane sugar factories (except in factories in Australia (1), as having the disadvantage of causing too much dilution of the sweetened juices and causing acetic acidification of these juices, would gain to be applied more in distillery, because it allows a much better extraction of the sugar.
(1) It was also released a few years ago under the Nobel process.

FIG. 5. – Simultaneous manufacture of sugar and alcohol.
The extraction rate, that is to say the amount of sugar existing in the extracted juice relative to the total sugar of the cane, is very variable depending on the facilities. In small distilleries, the percentage of bagasse sugar can rise to 10% and the extraction rate to be only 80-85%. On the contrary, when there are powerful mills, the extraction is often, because of better bagasse imbibition, as good as in the sugar factories (92-96%). Finally, if we practice maceration, it can reach, under excellent conditions, up to 98%.
The diluted juice from crushing of the cane flows into a bowl placed under the mills. It then goes on a bagasse sieve. In the small distilleries it consists of a simple perforated copper plate and, in larger plants, a long and narrow corridor arranged parallel to the mills. It has a horizontal portion carrying a copper cloth and an inclined portion terminating at the level of the intermediate conveyor of the second mill. Fine bagasse retained by the sieve is taken by an endless chain, with wooden squeegees, and is rejected after the first mill. The sieved juice is sent immediately to the composition pit, or, more often, directly to the fermentation tanks.
In small installations, the bagasse, after the last pressing, is received in a large basket that a worker takes to the generator or to the bagasse shed. The evacuation is carried out, in some cases by a mechanical transporter.
A. C. Amador (1) recommends the following process to simultaneously manufacture sugar and alcohol from the cane (FIG. 5):
(1) Fabricacion simultanea de azucar crudo y alcohol, in Almanaque 1945, 312. Ministerio de Agricultura, Cuba.
Juice from the shredder and the first 2 mills is pumped to scale C, where it is weighed and limed. It then goes into the juice heaters D, and is subjected to defecation, in the bins E. The clear juices are directed to the multi-effect feeding tray, while the sludge undergoes a second defecation in the trays I, which receive the previously heated juice of the last mills.
Clear juices of second defecation, after being cooled by passing through refrigerant M, are sent to the distillery, where they are subjected to fermentation, as is, or after mixing with molasses. The sludge of second defecation is received in the mixing tanks, diluted with hot water and used for bagasse imbibition, at the entrance of the fourth mill, if the installation comprises 5 mill trains.
This way of doing things makes it possible to reserve pure and concentrated juices for sugar, hence a very appreciable saving of fuel made in the evaporation of the juice, obtaining a sugar of better quality, reducing the amount of molasses, etc. …
Composition of the vesou.
Cane juice from the mill is in the form of a cloudy liquid, containing in solution sucrose, reducing sugars, gums, salts of organic and inorganic acids, dyestuffs, albuminoid materials, and a suspension of bagasse particles and soil. Among these substances, some are in the state of colloidal solution (proteic matters, gums, dyestuffs, free silica), the others in the state of true solution (sugar, proteoses, amino acids and mineral salts).
Composition is quite variable depending on the stage of maturity of the cane, the variety, the factors that have governed the vegetative development of the plant (soil type, fertilizer, climate) and the mode of extraction of the juice (mill pressure, hot or cold soaking, etc.).
Honig (2) indicates the following percentage of cane reaching full maturity and the distribution rate of the various constituents between the juice and the bagasse:
(2) Arch. Suikerind. Ned. Indië XLII. 249. 1934.

The normal cane juice coming from the mills would, according to Hardy, have the following composition (by weight):
Eau … 80 – 85 %
Saccharose … 10 – 18 –
Reducing sugars 0.3 – 3.0 –
Organic Matter 0.5 – 1.0 –
Mineral Matter 0.2 – 0.5 –
The average centesimal composition of the non-sugar would be, according to the same author:

Before examining the various constituents of juices, we reproduce below some analyzes, which illustrate variations of composition of the product under the action of various factors.
Honig (1), in Java, reproduces among other analyzes relating to the composition of vesou at different stages of plant development, the following, made in 1931 on canes from the district of Modjo:

McKaig and Fort (1), in Louisiana, found as composition juices (juice of the shredder) of healthy canes and canes damaged by the borer (Diatraea saccharalis F.) and by the fungus of the red snot (Colletotrichum falcatum Went):
(1) J. Agr. Res. LII, 17, 1936.

The non-sugar of the previous canes contained % dry matter:
(2) In cc of 0.1N soda required to neutralize 10 cc of juice (titration of phenolphthalein).
Density, sugars.
The density of cane juice is subject to significant variations depending on saccharine richness of the plant and the rate of imbibition. It oscillates in general, for undiluted juice (juice of the shredder or the first mill), between 1.060 and 1.080. But we have seen where mixed juice density can be lowered, in agricultural distilleries, to around 1.040, by the use of a large quantity of water for imbibition.
Brix of the undiluted juice, i.e., the amount of dry matter p. 100 of the juice by weight, usually varies between 14 and 20 (16-18 by the average weight of salts dissolved in the vesou being higher than that of the sucrose solutions of the same concentration, the apparent dry extract given by the Brix saccharometer is greater than the actual dry extract obtained by liquid evaporation. According to Fort and McKaig, we can calculate this difference by multiplying the apparent Brix increased by percentage of carbonate ash by the factor 0.0120.
Cane juice normally contains only 3 sugars: sucrose, glucose and levulose. The latter two (reducing sugars), which represent intermediate bodies in the synthesis of sucrose, gradually diminish as the plant approaches maturity. In the juice of ripe canes, their proportions usually oscillate between 0.3 and 2%, for sucrose levels of between 12 and 18% (14-16 on average).
If the cane is very far from the point of maturity, glucose and levulose exist in approximately equal amounts. However, when maturity is advancing, levulose is used more rapidly than glucose and tends to disappear (Prinsen-Geerligs, Browne and Blouin). In some rare cases, glucose itself can be in turn fully used, so that the cane contains only sucrose, no reducing sugars (Spencer).
In canes having exceeded their point of maturity, attacked by rats or insects, burned or left to their own after being cut, sucrose inversion occurs more or less rapidly, under the influence of certain microorganisms or by invertase, which normally exists in the tops of the cane and diffuses, after cutting, throughout the stem.
These different causes explain that the coefficient of purity of the juice, that is to say the percentage of sucrose relative to the total dry matter, is subject to great variations. In regions bordering the growing zone (Louisiana, Argentina), where the cane is never perfectly ripe at the time of harvest, and in equatorial countries (English Guyana), where clumps of cane contain stems at all stages of maturity, the average purity hardly exceeds 80 and can descend at the beginning of the season to 70. On the contrary, when climatic factors allow good plant maturation, the purity reaches 83-85 and even in favorable cases (canes of dry regions, cultivated under irrigation) 90 and more.
The apparent purity, obtained from the apparent Brix and directly polarized sucrose, is always lower (usually 4 to 6%) than the actual purity, calculated from the actual dry matter and sucrose determined by double polarization.
Acidity.
Vesou has an acid reaction. In the case of mature and healthy canes, this acidity is low: it is generally between 0.6 and 1.6 grams per liter, as acetic acid (1).
(1) It is more appropriate to evaluate the acidity in acetic acid than in sulfuric acid, the average normal weight of the organic acids in the composition of the vesou being approximately 60, normal weight of acetic acid.
The concentration of the hydrogen ion juice is approximately constant, vesou having a high buffering capacity. Paine and Bach (2) observed in Porto Rico that the pH of undiluted vesou ranged from 4.73 to 5.29 (5.09 on average); Gomez (1), in the Philipines, found an average of 5.39 (5.26 to 5.57); Fort and McKaig, Louisiana, 5.38.
(2) Facts ab. Sugar XXII, 338, 1927.
(1) Philippine Agr. XIX, 609, 1981,
Canes burned before cutting, left to themselves for some time before being handled or parasitized have a much more acidic juice. The pH of the vesou from canes strongly affected by insects or fungi goes down to 4.0 – 3.8 (Iwata).
Similarly, the expressed juice acidifies rapidly, even before having undergone a beginning of alcoholic fermentation: after 24 hours, the acidity can reach 30 grams in SO4H2 per liter. This “souring” comes from the action of bacteria, which seem to directly attack sugars and which give rise to volatile acids (among which predominates acetic acid) and especially to fixed acids (Tempany).
The acidity of the vesou is due to the presence of acidic mineral salts (sulphates and phosphates) and organic acids (aconitic acid, malic acid, oxalic acid, etc.).
Yoder (2), in Louisiana, found per liter of cane juice: 0.50 grams of aconitic acid, 0.0077 grams of malic acid and 0.0004 grams of oxalic acid. He did not meet tartaric, citric or succinic acids.
(2) Ind. Eng. Chem. III, 640, 1911.
Mc Allip (3) observed that the level of aconitic acid, which is a dehydrated citric acid and has the formula CO2H – OH = C (CO2H) – CH2 – CO2H. could vary in fairly large proportions, depending on cane variety and locality. He found in various syrups, prepared without defecation with lime, rates ranging between 0.880 and 1.330 gr. of aconitic acid per 100 gr. dry matter.
(3) Ind. Eng. Chem. XXXIII, 637, 1941.
Balch, Broeg, and Ambier (4) found that the proportion of aconitic acid in the blended juices of Louisiana sugar factories ranged from 0.15 to 0.31% of the juice. It is 3 to 5 times higher in juices from immature ends (white tips) than in those provided by the body of the cane. The level of aconitic acid also depends on cane variety and the nature of the crop soil. In general, it is roughly proportional to the total acidity of the juice.
(4) Sugar XL, No. 10, 32. 1945.
Tanaba evaluated cane juice from Formosa, in gr. per liter of vesou:
Acide aconitique 0.119
>> malique 0.0063
>> citrique 0.0006
>> oxalique traces
He found no trace of tartaric, lactic, succinic and fumaric acids.
Unlike previous authors and several others (Behr (5), Taylor (6), etc.) who observed that the dominant organic acid of cane juice was aconitic acid, Winter and Prinsen-Geerligs (7). have been able to detect the presence of this body in Java juices and syrups. On the other hand, the canes of this country are richer than those of Louisiana in oxalic acid: Bosz (8) found 0.014% in the P.O.J. 100.
[P.O.J. is a cane variety]
(5) Proc. Amer. Chem. Soc. 1876, 220. Arno Behr is the first author who reported the presence of aconitic acid in cane juice.
(6) J. Chem. Soc. (Trans) CXV, 886, 1919.
(7) Arch. Sulkerind, Ned. Indie XXXI, 720, 1923.
(8) Arch. Suikerind. Ned. Indie XXVIII, 969, 1920.
Acetic acid is found in appreciable quantity in vesou only if the cane was previously damaged by fire, jelly, etc… or if the juice has undergone a beginning of fermentation.
Mineral matter.
The mineral content of cane juice is subject to great variations. Pitcairn (1) has found, in the Hawaiian Islands, 0.29 to 0.62% ash juice: Spencer, Cuba, 0.25 to 0.60; King (2) in the Philippines, 0.31 to 0.63% (mixed juice); Fort and McKaig in Louisiana, 0.40 to 0.72% (shredder juice), corresponding to 2.63 – 4.01% dry matter.
(1) Facts ab. Sugar X, 128, 1920.
(2) Ind. Eng. Chem. XXIII, 954. 1931
Agrological conditions (soil, climate, manure, etc.) obviously have a great influence on mineral matter richness. The varietal nature of the plant is also involved. Coates, Fieger and Salazar (3), for example, have observed that the varieties formerly grown in Louisiana (Louisiana Purple, Louisiana Stroped, D. 74) contained an average of 3.1% ash as dry matter in the cane, while P.O.J. 36, 213 and 234 contained 3.9%, an increase of 22%. The amount of ash from juices also increases with the pressure of the mills (Prinsen-Geerlig, Thieme). However, according to Thieme (4), the most important factor would be soil moisture: the relatively dry regions produce high ash canes, and this rate is also higher in dry years than in rainy years.
(3) Plant. Sug. Manuf. LXXX, 422, 1928.
(4). Arch. Suikerind. Ned. Indië XXXVIII, 1155, 1930.
The main mineral constituents of cane juice are silicon, calcium, magnesium, potassium, sodium, phosphorus, chlorine and sulfur. Other less important elements are iron, aluminum and manganese.
Like that of total ash, the proportion of the different elements undergoes important variations.
Spencer gives the following centesimal composition of the ashes of juices obtained in 1912, in different factories of Cuba:
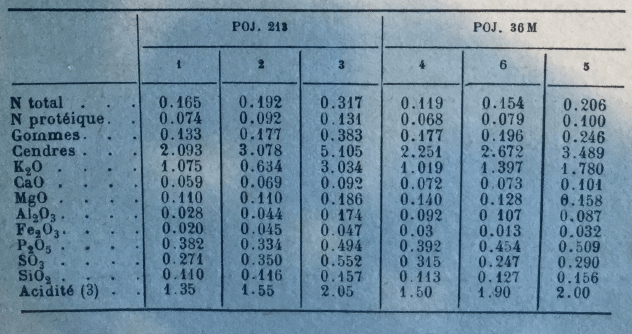
 Fort and McKaig found the following average minima and maxima for various varieties of Louisiana cane (POJ 213, 234, 36 – M, Co 281, 290, CP 807, 28 – 19, 28 – 11), harvested in 1931 and 1932.
Fort and McKaig found the following average minima and maxima for various varieties of Louisiana cane (POJ 213, 234, 36 – M, Co 281, 290, CP 807, 28 – 19, 28 – 11), harvested in 1931 and 1932.

Average ash rates varied between 2.63 and 4.01% dry matter juice.
Saline materials are in the form of mineral salts (phosphates, sulphates, chlorides) or organic salts. The organic acid radicals being heavier than the mineral radicals (carbonates or oxides) evaluated in the ash, it follows that the ash content is lower than that of the salts actually present.
Fort and McKaig calculated the content of Louisiana juice in mineral salts and organic salts (mg per 100 cc of juice from the shredder). Phosphates and sulphates have been evaluated as acid salts, chlorides and organic salts as neutral salts, organic acid radicals being generally monobasic (Shorey).

According to the above results, total salt content is thus 58 to 72% higher than that of ash, while the proportion of organic salts relative to total salts varies from 54.2 to 62.5%.
Silica is found only in small proportions in the vesou. It seems to come mainly from soil adhering to the cane and fine bagasse particles, rather than the sap itself. It exists in the form of silicates and especially of colloidal silica in the free state or combined with other colloids. According to some authors, it would also be in the form of esters, in combination with certain polysaccharides.
Phosphoric acid, which is in the form of organic and inorganic phosphates, passes almost completely into the juice during cane crushing (Honig, Saint (1) Some varieties are much richer than others in this element.
(1) Prec. 5. Cong. Int. Soc. Sugar cane Techn., 616, 1936.
Sulphates generally exist in about the same proportions as phosphates, but they are subject to greater variations. These depend mainly on the soil’s richness in sulphates and chemical fertilizers.
Of the different mineral elements of vesou, potash is the most important: 25 to 50% of the total ash. The extraction rate of potash from the cane is about the same as that of phosphoric acid and sucrose (Honig, Saint).
Soda ash is found in much smaller quantities: the soda-potash ratio averages 1/25 in cane juice (Fort and McKaig), while in beet juice it reaches 1/3.
The proportion of alumina, which comes, at least in part, from the soil adhering to the canes, is nearly the same as that of soda and iron. As for manganese, it exists only in very small proportions.
Nitrogenous substances.
Nitrogen is found in cane juice in the form of proteinaceous matter (proteins proper and nucleoproteins), coagulable by heat; proteoses (albumoses and peptones), products of hydrolytic degradation of the preceding materials, not coagulable by heat; amino acids and amides; and finally mineral materials (nitrates and ammoniacal salts). Proteins and proteoses are in colloidal solution, the others in true solution.
The amount of nitrogen compounds in the juice, generally estimated by multiplying the nitrogen by 6.25 is highly variable, Fort and McKaig, in Louisiana, obtained, for the individual samples of vesou (juice of the shredder), extremes of 0.294. and 2.488 (0.590 and 1.057 on average) % dry matter. Protein nitrogen relative to total nitrogen is also variable: 18-81% (average 50-56) in Louisiana juices, Fort and McKaig: 75-90% in Hawaii, according to Shorey.
Ecological conditions, the stage of cane maturity and varietal are involved in these variations. The amount of nitrogenous material passing through the juice can vary from 40 to 60% according to Honig, the pressure exerted by the mill and the rate of imbibition are also taken into account. Canes harvested before maturity, as well as those attacked by parasites, the tops of the stems (“white tips”) give a juice richer in nitrogenous materials and fermenting more easily than that of normal canes (Iwata). The nitrogen richness of vesou also increases with the pressure of the mills: Fort and McKaig, for example, found in the juice of the shredder 0.064 of protein nitrogen and 0.056 of non-proteic nitrogen % of dry matter versus 0.073 and 0.088 % in the mixed juice.
The nature of the nitrogenous organic compounds of the vesou is still poorly known. Shorey (1) has found as amino acid glycine or glycine (aminoacetic acid) NH2 – CH2 – CO2H. According to Zerban (2), who could not identify the previous body, the main amino acid would be aspartic acid, or aminosuccinic acid CO2H – CH2-CH NH2) – CO2H. This acid is mainly in the form of an amide, asparagine NH2 – CO – CH2 – CH (NH2) – CO2H, which is particularly abundant in the juice of unripe canes. The same author also isolated glutamine, the amide of aminoglutamic acid having the formula NH2-CO-CH2-CH-CH (NH2) -CO2H, and another amino acid, tyrosine, or oxyphenylamino-propionic acid. But these last two bodies are in much smaller proportions than asparagine.
(1) J. Amer. Chem. Soc. XIX, 881. 1897 : X, 133, 1898 ; XXI, 609, 1899.
(2) Proc. 8. Int. Cong, Appl Chem. 1912.
Shorey has recognized the presence in vesou of various lecithins, distearic phospahtides in one of the acidic functions of phosphoric acid is saturated with a nitrogenous base. The bases identified by the author are choline (CH2) 3 ≡ N (OH) – CH2 – CH2OH, and betaine CH2 – N (CO2) ≡ (CH3)3, an oxidation product of choline. Guanine, amide of the purine series, has also been identified. In 6 samples of vesou studied by the author, guanic nitrogen content was 0.0012% and that of the total nitrogen 0.0350%.
[SOS there may be a typo in that last chemical formula
Reader, Jérôme, is helpful to clarify (We now know we can ask him complicated chemistry questions!):
Choline is (CH*3*) 3 ≡ N (OH) – CH2 – CH2OH,
betaine is CH2 – N (CO2) ≡ (CH3)*3*
(of course without highlighting “*”).
Three things:
-I don’t want to be picky, but numerals should be in indices (but maybe they can’t due to blog’s limitations),
-it’s an old way to picture molecules, in order to show molecules in text-only, and thus have many limitations (and some benefits altogether),
-(OH) (in fact OH- , with the “-” being in superscript) in choline is a counter-ion in order to compensate the ammonium cationic charge as we show it today. There is also an ammonium in betaïne, but the counter-ion is the carboxyl (COO-) at the end of the molecule (the molecule being a zwitterion). So, it should be shown as:
carboxylate(-)-methylene-ammonium(+)≡methyls
but the writer had the carboxylate “glued” to the ammonium it compensate:
methylene-ammonium(carboxylate)≡methyls
and the trap is that methylene has in fact “one arm” on ammonium and “the other arm” on carboxylate, but the formula shows only one bond, just because showing two between methylene and ammonium is incorrect, leading to the fact that “gluing” ammonium and carboxylate facilitates the understanding that they compensate each other in term of ionic charge, but make writing the formula tricky…]
Mineral nitrogen exists only in very small proportions in cane juice. Ammonia salts are only found in trace amounts.
Gums.
Organic substances of the alcohol-precipitable organic matter are commonly referred to as “gums”. These include not only true gums, which are soluble in water and give reducing sugars by hydrolysis, but also various hemi-celluloses (lignin, etc.).
The amount of gum existing in cane juice is very variable. It increases with the pressure of the mills and the temperature of the water mixture (Browne and Blouin). Cane variety, plant maturity and the time that elapses between cutting and milling also influence it. The proportion of gums is higher in young canes and those which are left to themselves for some time before being handled (Steuerwald). Some parasitic diseases, such as gummosis, caused by Bacterium vascularum Cobb, strongly increase the amount of gums: the juices are thick and viscous, very difficult to work in the sugar factory.
Gums are derived from cane hemi-celluloses and are formed mainly by pentosans: xylan and araban, with small amounts of galactan (immature canes). However, those of parasitic origin consist of dextran (gummosis) or levulan (Bacillus levaniformans). Some of the gums are in the form of pectins.
Steuerwald and Hazewinkel (1) found that Java mixed juices contained 0.25 to 0.38% pentosans dry matter, with those from the first mill containing only traces and those from the last mills from 0.77 to 1.21%. Farnell (2) measured 0.14 to 0.34 pentosanes and 0.08 to 0.38 pectins (lime pectates) % dry matter in mixed juices from Natal and Mauritius. The same author found that the proportion of pentosans and pectins was very low, (0.036 to 0.049% pentosan) in juices coming from cane crushing by a low-pressure apparatus (laboratory mills). Browne and Philipps (3), based on the proportions of methoxyl and uronic acid found by them, think that the gums of cane juices and molasses consist mainly of pectins.
(1) Arch. Suikerind. Ned. — Indië XIX, 313, 1913.
(2) Int. Sug. J. XXVI. 480. 1924: XXVII, 254, 1926.
(3) Int. Sug. J. XLI. 430, 1939.
Wax and fat.
According to Honig, 20 to 40% of the waxy materials that cover the surface of the cane goes into the juice. It contains quantities of wax very variable depending on the rate of extraction and especially the variety of cane considered, some kinds like the “Uba” having up to 0.05% wax and others almost no.
According to Wijnberg (4), 70% of raw cane wax is composed of the glycerides of oleic, linolic, palmitic and stearic acids, with resinous acids, lecithins, phytosterol, dyestuffs, and aromatics. The remaining 30% is represented by myricilic alcohol (about 45%) and another non-primary alcohol.
(4) J. Soc. Chem. Ind. 1912, 991.
A wax sample extracted from a Java sugar factory press sludge and examined by Bosz (5) had a melting point of 60-62° C., an acid number of 47.3 and a saponification number of 177. The fatty acids extracted by saponification, addition of hydrochloric acid and stirring with benzine, had a melting point of 54° C and appeared to be constituted by a eutectic mixture of palmitic and stearic acids. In the liquid obtained by steam distilling the product of the saponification, the presence of myricilic alcohol and caproic and formic acids was recognized. Acetic, benzoic and cinnamic acids and cholesterol could not be detected.
(5) Arch. Suikerind. Ned. Indie XXVIII, 974, 1920.
Coloring matters
The main coloring substances in cane juice are chlorophyll, anthocyanin, saccharetin and tannins.
Chlorophyll from the cells that passes into the juice remains in colloidal suspension and is eliminated during defecation; it is more abundant in the juice of unripe canes.
Anthocyanin is a purple pigment found in epidermal cells of red or purplish cane varieties. Soluble in water, it becomes dirty green in alkaline medium and dark brown in the presence of ferric salts.
Dasa Rao, Walawalkar and Strikanton (1) were able to isolate the coloring matter from Mauritius “purple cane” and found that it consisted of a diglucoside of formula C28H33O15Cl. H2O. maybe ampelopsidine.
(1) J. Indian Chem. Soc. XV, 27, 1944.
Steuerwald (2) has given the name of saccharetin to an ill-defined material which permeates cane fibers; colorless in acidic medium, it takes a dark yellow tint in alkaline medium and dark brown in the presence of ferric salts. It is introduced into the juice by fine bagasse particles in suspension and dissolves during liming.
(2) Int. Sug. J. XIV, 153, 1912,
Finally, we find in the tops and eyes of the cane polyphenols, or tannins, which pass partly in the juice and take contact with the iron mills forming a dark green coloring. Among these polyphenols, according to Zerban (3), we find a true tannin derived from pyrocatechol and related to oak tannin.
(3) Ind. Eng. Chem. X, 814, 1918; XI, 1034, 1919, XII, 744, 1920
According to the same author, the dirty green color of vesou results mainly from the interaction of polyphenols and ferrous salts formed by the action of organic acids in the juice on the iron of the rollers and then transformed into ferric salts by oxidases in the cane. The brownish color may also appear in the absence of iron salts, but it is then 2 or 3 times less pronounced. It is due, in this case, to the action of a laccase on the polyphenols and, for a small part, to that of a tyrosinase on the tyrosines of the juice.
Besides the substances which we have just indicated as being part of the composition of vesou, there are others, not yet determined, which may play an important part in the constitution of the bouquet of eaux-de-vie. Of particular note is an aromatic principle that communicates to cane juice a particular taste, called “taste vesouté”. This principle is found in rum and can only be eliminated by defecation and concentration of the juice.
Value as raw material of fermentation.
Cane juice, whose composition is close to that of grape juice, except for less acidity, is an ideal medium for fermentation.
It also contains a high number of microorganisms. Owen found in Louisiana 250,000 per cc of raw juice; Nakazawa, Takeda and Suematu, at Formosa, from 104,000 to 1,000,000 per cc. These microorganisms, which include yeasts proper (Saccharomyces and Schizosaccharomyces), Torula, Monilia, mold, acetic, butyric, lactic, putrefactive bacteria, etc., are provided by the cane itself (on the surface of which they live), by the earthy matters which defile it or by the air. Abandoned to itself, the juice immediately enters fermentation.
Browne, however, reported that the newly extracted vesou has germicidal properties and that during the first days after extraction, the number of microorganisms decreases. This is no longer the case when the canes have been damaged by frost or fire: in this case, deterioration of juices continues rapidly as soon as extraction is done.
In order to achieve optimum conditions for alcoholic fermentation, it is advisable to lower the concentration of vesou to about 1.040-1.050 and to increase the acidity by the addition of a mineral acid. The juice provided by ripe canes is also insufficiently rich in nitrogen and sometimes in certain mineral salts (phosphates). Alcoholic fermentation is consequently improved by the use of a small quantity of sulphate of ammonia. On the other hand, the juice of immature canes, damaged by parasites and those coming from the tips (white tips), do not need nutritive salts added, because they have a higher content of nitrogen and mineral matter (Iwata).
Ohtsu also reports the presence in the tops of the cane of a crystalloid substance that stimulates yeast activity. By adding 0.008 of this substance to over ripe cane juice, he has achieved a 40% increase in fermentation activity and a sharp reduction in fermentation duration. In the treated juice, yeast multiplied actively and had larger cells. [What the hell is happening here? Fascinating]
Molasses
Molasses, which is the raw material most used in rum manufacture is the residual syrup of sugar manufacture.
Depending on whether it comes from the centrifuging the sugars of the 1, 2 or 3rd centrifuge, it is called first, second or third molasses. More often, in French, the term “molasses” is reserved for the final product, of which it does not belong to sugar. It is no longer possible to extract more sugar economically, the intermediate syrups are rather referred to as “égouts” [sewage]. In Cuba and Louisiana, the name “blackstrap” is used to describe molasses sold from the manufacture of 96° polar sugar.
It is especially final molasses which is used in distillery, the first or second molasses, when they do not enter the manufacture, being rather reserved for the direct culinary consumption (United States). However, in Jamaica, first-order molasses are normally directed to the distillery.
In addition to sugar, molasses contains impurities in the raw juice which could not be eliminated by the purification work; products formed by the decomposition of certain elements of non-primary sugar, in the presence of the reagents used for purification; finally bodies resulting from the alteration of sugar by heat and lime. Its composition is very complex and varies quantitatively with the nature of the canes implemented and conditions of work in various phases of manufacture. Methods of purifying the juice (defecation of the skin, sulphitation or carbonation) have a particular influence on the composition of non-sugar.
[This is where rum oil begins. Molasses can also be thought of as an investment product and not merely a byproduct.]
We give below some analyzes of final molasses coming from different sugar producing countries.
Java Molasses (Prinsen-Geerligs)

Molasses 8 and 9 are molasses of carbonation, others molasses of defecation.
Molasses from Hawaii Islands (Peck and Deer)


Here is the comparative composition of molasses of different centrifuges obtained in Louisiana (by Browne) (1):
(1) Louisiana Exp. Sta. Bull. No. 93 1907.

Reich (1) gives the following composition for various types of molasses:
(1) Molasses distillers; dried yeast. Sugar XL No 3, 26-29, 1945.

Finally, Arroyo found for molasses from Puerto Rico:

Sugars.
Unlike beet molasses, which contains an average of 45% sucrose and have no or very little sugar reducers, cane molasses, which usually ranges in density from 80 to 90° Brix (1.412 to 1.480), contain large quantities of reducing agents.
The proportion of these is very variable. It depends mainly on the amount of reducing sugars existing in the cane. The methods of making sugar are also involved: if one works in an acidic medium (manufacture of white sugar), there is a inversion of sucrose, which does not take place in neutral or slightly alkaline medium.
The amount of reducing sugars is sometimes reduced to below 10% in the case of mature cane molasses with a low glucose content. On the contrary, it may exceed 35% when the canes have not reached maturity or have undergone a beginning of alteration. It is on average 20 – 25%.
The proportion of sucrose is all the higher as that of the reducing sugars is lower, at least in the case of depleted molasses (1). It also varies between 30 and 40%.
(1) When the production of rum is lucrative, we generally tend not to exhaust molasses, whose composition is then intermediate between that of syrups and molasses (Jamaica, French West Indies).
It is generally accepted that a molasses can be considered as commercially exhausted, when the apparent purity, Pol x 100: apparent brix, is close to 30, and absolute or real purity, Sacch real x 100: Real Brix, close to 40. The apparent purity, however, provides only vague indications because the ratio between polarization and sucrose is extremely variable, optical activity of the reducing sugars being very low or even nil in some molasses (molasses of carbonation of Java in particular) and high in others. Molasses purity depends essentially on the ratio Reducers : Ashes, according to Prinsen-Geerligs, and is as great as this ratio is weak.
Cane molasses reducers consist essentially of glucose and levulose, in proportions that are not equal as in the invert sugar, but unequal and variable according to the quality of the cane used. In addition, there are small amounts of mannose and non-fermentable reducers which have been referred to as glutose. Mannose has been reported especially in Egyptian molasses by Pellet (2). It would average 0.4%, according to L. de Bruyn and A. van Eckeinstein.
(2) Pellet also reports the presence of raffinose in Egyptian molasses. But this assertion could not be confirmed by the other authors.
Glutose. — Lobry de Bruyn and Alberda van Eckeinstein (3) observed, as early as 1897, that monoses (glucose, levulose, mannose) undergo, in the presence of alkaline or alkaline-earth bases, a transformation by which a small quantity of a non-fermentable reducer hexose, which they called “glutose”.
(3) Rec. Trav. Chim. Pays-Bas XVI, 274, 1897.
Many authors have identified with this sugar the nonfermentable reducing material found in cane molasses of various origins. We give below some figures relating to the proportions of these reducers (calculated in glucose % of molasses):

Note that beet molasses contain at most 1% of infermentable reducers.
Colthof (4), repeating the experiments of L. de Bruyn and A. van Eckeistein, was not able to obtain by treating the fermentable monoses by bases a unique and well-defined reductive substance. He therefore considers that “glutose” is, not a specific chemical substance, but a mixture of substances of unknown constitution and probably very variable, including hexoses and bioses, glucic acid, saccharinic acid, etc. These conclusions were confirmed by Reindel and Frey (5), who were able to prepare two different osazones from the unfermentable residue of a Natal molasses. These authors also showed that unfermentability could be partly due to the presence of inhibiting substances and to a high ash content.
(4) Biochem, Z. 191, 1931.
(5) Z. Spiritusind. LVII, 237, 1934
According to Sattler and Zerban (1), the glutose of Lobry de Bruyn and Alberda van Eckeinstein is constituted by a mixture of fructopyranose anhydride and difructopyranose anhydride, with small quantities of other reducing substances: complex anhydrides of glucose, comparable to caramel, hydroxymethylfurfurol, d-allulose (encountered by the above authors in the “glutose” of cane molasses), etc.
(1) The nature of glutose. Sugar XXXIX, No. 12, 28, 1944.
Benedict, Dakin and West have observed that glutose is chemically inert in the human body and is excreted without transformation. Yeast added to a mixture of glutose and sodium phosphate can not form hexosephosphate.
Infermentable reducers of cane molasses result mainly from the action of lime on glucose and especially levulose, during defecation of the juice. A series of decomposition products having a high molecular weight and different chemical properties are formed. Some of these may undergo fermentation after hydrolysis with dilute acids; others become optically active, while others are finally transformed into insoluble humic substances. It is mainly the duration of action and the concentration of lime which affect the quantity of infermentable reducers (Müller) (2). According to Waterman and van der Ent (3), these, which would already exist in newly expressed cane juice, would increase during juice purification, but not during evaporation and cooking. Their formation is facilitated by the rise in temperature and pH. The rate of infermentable reducers also increases during the conservation of molasses during the first months after manufacture (La Bastide).
(2) Arch, Suikerind. Ned Indie XXVI, 346, 1918.
(3) Arch. Suikerind. Ned. Indië XXXIV, 943, 1926.
Acidity.
Molasses has a slightly acidic, neutral or slightly alkaline reaction. Most often, they show 3-6 grams of acidity as sulfuric acid per liter. Their pH varies between 4.5 and 7.5 (5-6 on average) Molasses obtained from the manufacture of white sugar are acidic, and those derived from the manufacture of brown sugar with 96° polarization are slightly acidic, neutral or alkaline.
The combined acids are mainly in the form of salts of potash, lime and magnesia. The principal mineral acids are hydrochloric, sulfuric (4), carbonic, phosphoric and nitric acids.
(4) Sulfur is also found in molasses which have been purified by sulphitation, under strong sulphites (Ca, K, Na). These can undergo reduction during fermentation giving rise to H2S [hydrogen sulfide], or decomposition with release of SO2 [sulfur di-oxide]. These gases combine with alcohol to form ethyl sulphide and sulphite, of very unpleasant odor which can depreciate the obtained eau-de-vie. The smell of hydrogen sulphide is sometimes very noticeable in juices and syrups that have been left standing for a long time.
Nelson found in a molasses of Cuba the following organic acids:

In addition, there are amino acids (aspertic acid and glutamic acid) and dark-colored, uncristallizable acids of poorly known constitution: glucic acid, glycolic acid, melassinic acid, saccharinic acid.
Balsh, Broeg and Ambler (5) found in blackstrap molasses from different origins (Cuba, Louisiana, etc.) variable and often very high proportions of aconitic acid (0.96 & 6.13 dry matter). Part of the acid is in the form of an insoluble compound: the sediments that come from centrifugation of diluted molasses contain in fact from 16 to 35% of aconitic acid. Variability of the level of this element in molasses undoubtedly depends on the original content of cane in aconitic acid, which is eliminated in small proportions during manufacture of sugar and passes almost entirely in the molasses.
[This is an interesting note as more people explore molasses pre-treatment with centrifuges.]
(5) Sugar XL. No. 10, 32, 1945.
Takei and Imati have isolated acetic, butyric and benzoic acids from the ether extract of Formosa molasses, as well as another unidentified volatile acid with the characteristic fragrance of molasses. By treatment with dilute sodium hydroxide, this last aromatic material was completely destroyed. In the neutral portion of the ether extract, the same authors found stigmasterol C29H48O and syringic acid or ac. dimethylgallic, of formula (CH3-O)2 = C6H2 (OH) 2 – CO2H5.
Glucose, glycolic, melassinic and saccharinic acids result from the action of lime on glucose and levulose during juice purification. They are very unstable. Glucic acid, for example, which has the formula, according to Nelson and Browne (1), CHOH = CH – CO2H (β-hydroxyacryylic acid), is easily oxidized on contact with air, giving formic and acetic acids. The level of formic acid can reach a significant figure in some cases: up to 0.154% in Cuban molasses and 0.791% in those of refinery, according to Zerban (2).
(1) J. Amer. Chem. Soc. LI, 830, 1929
(2) J. Ass. Off. Agr. Chem. XV, 355, 1932,
Acetic and lactic acid also come from the action of lime on sugars, while aconitic, malic and citric acids pre-exist in cane juice.
Finally, during molasses storage, bacteria and mold infections can occur, giving rise to various acids such as oxalic acid, citric acid and butyric acid.
Mineral matter.
The mineral matter content of final molasses is generally 8-10% (10 to 12% dry matter). In dry countries it can rise to 15% and lower when the molasses is poorly exhausted, at 5%.
The saline concentration of the molasses is therefore on average 3 or 4 times higher than that of the vesou (2 to 4% of dry matter). The balance of mineral elements is also significantly different in these products. During juice purification, certain elements are, in fact, eliminated for the most part: this is the case for phosphoric acid, silica, alumina, iron and manganese, of which more than 80% are precipitated. Others are not reached and pass entirely in molasses: potash, soda, hydrochloric acid. Others finally increase in absolute value, by the contribution of the reagents used: lime and, in the case of purification by sulphitation, sulfuric acid. By way of example, we reproduce below results obtained by Fort, (3), in Louisiana, by clarifying at pH 7 juices mixed by the processes of defecation and suifitation:
(3) Facts ab. Sugar. XXIX sept. 1939, 34.

The mineral composition of molasses is very variable. Here are some analyzes of the ashes of molasses from various countries (% of ash).

Prinsen-Geerligs found for various Molasses of Java (% molasses):

Nitrogenous substances.
Nitrogenous materials (N x 6.25) of the molasses generally vary between 1.5 and 5%.
It consists mainly of amino acids (aspartic acid, glutamic acid, etc.) resulting from the hydrolysis of vesou nucleoproteins and transformation of amides into amino acids under the action of lime (asparagine is especially transformed into aspartic acid). During juice purification, most albuminoid material is coagulated and eliminated; the other nitrogenous principles are little or not affected. Farnell (1) found in Natal molasses, 0.054% colloidal nitrogenous material (albuminoids) for 1.32 to 2.52% total nitrogenous matter, and in maize molasses: 0.048 to 0.198 of colloidal nitrogenous material, for 2.11 to 4.88 % of total nitrogenous matter. The ratio of albuminoid nitrogen to total nitrogen varied widely, depending on the samples (1 to 12%).
(1) Int. Sug. J. XXVII, 254, 1925.
A small amount of nitrogen is also in mineral form: N ammonical (traces) and N nitric. Pellet and Müller (2) measured 0.47% potassium nitrate in the molasses of Egypt, corresponding to 0.02% of handled canes.
(2) Int. Sug. J. XIII, 493, 1911.
Coloring materials and waxes.
Chlorophyll is eliminated during defecation. Other dyestuffs in the juices (anthocyanin, saccharetin, tannin) undergo various transformations: the process of sulphitation removes a greater quantity than the defecation with lime. Finally, during manufacture, by the action of lime on the sugars, staining materials are formed (comparable to those which originate in the preparation of the caramel), which give molasses a more or less pronounced brown color.
Liming removes most of the wax and fat from the vesou, which ends up in the sludge. A certain quantity, however, passes into the molasses, while another is deposited in the form of calcium soaps in the tubes of the evaporation cases (incrustations). Farnell found 0.1. 026% wax in molasses from Natal and Trinidad.
Gums and Various.
Pentosans and pectic substances of the juices escape for the most part the action of the purifying agents. Sulphitation and carbonation would eliminate a certain quantity, but liming eliminates very little. Farnell found from 0.14 to 0 46% pentosanes in molasses from Natal and Mauritius. In 3 molasses from Trinidad, the same author measured 0.55 – 0.72 pentosanes and 0.33 – 0.70 pectines (in pectate lime) % Brix. According to Browne, molasses gums are largely composed of pectins.
Pentosans usually found in molasses are araban and xylan. Other substances which may be accidentally encountered include dextran, levulan, cellulan, mannan and galactan.
Dextran appears quite often in geleed canes or those attacked by insects (Browne), the juice having undergone gummy fermentation (Leuconostoc), etc. Due to its high rotatory power (+ 200 °), it introduces an important cause of error in the polarization of juices and molasses.
Levulan results from the activity of various bacteria, including Baillus levaniformans. It provides, by hydrolysis with acids, levulose, while dextran gives glucose.
Cellulan was found by Browne in Louisiana. It is formed in certain fermentations of juices and molasses. It is in the form of large membranous lumps, insoluble in caustic soda (which differentiates it from dextran). When treated with hot soda, it gives a product presenting the reactions of cellulose, and, hydrolyzed by acids, it transforms into glucose.
Mannan is sometimes found in sedimentary deposits of fermented juices and molasses. It gives mannose by hydrolysis with acids.
Galactan or galacto-xlyane gum was found in significant quantities by Gupta, Sen and Watson (1) in molasses obtained in the manufacture of gur (brown sugar produced in India by indigenous methods). It is very resistant to hydrolysis by acids and provides by oxidation a large amount of mucic acid. It has pronounced reducing properties and consequently introduces a cause of error in the analysis of reducing sugars. It is to this cause that the above authors attribute the defective alcoholic yield of molasses examined by them. Galactan does not normally exist in molasses (Farnell).
(1) J. Soc. Chem. Ind. XLIII, 291, 1924.
Finally, Browne has encountered relatively high amounts of chitin in the froths that form on the surface of Louisiana molasses kept in bins. This body, which appears to have a fungal origin, gives by hydrolysis with HCl, glucosamine C6H13NO5.
These scums also contain a significant proportion of a fat whose composition is similar to that of cow butter. However, it is more acidic than fresh butter and has a predominance of soluble acids (caproic and caprylic acid).
A sample of scums offered, after removal of the molasses, the following percentage composition: water 10, chitin 11.30, pectin 31.61, fat 27.50, ash 5.58, undetermined 14. During distillation, the fat decomposes giving acrolein and other undesirable products that spoil the resulting eau-de-vie. It is true that the inferior quality of rums supplied by certain types of molasses is due to the presence of fat in them (Owen). Elimination of the foam that forms on the surface of the molasses during their storage is therefore advisable.
We often find in molasses a hexatomic alcohol mannile, CH2OH (CH OH4 – CH2OH) which is formed in various fermentations, and in particular in that caused by Leuconostoc, by reduction of the invert sugar. The quantity of mannite varies moreover rapidly, this product being attacked by many bacteria.
Conservation of molasses.
The storage and handling of molasses was formerly done in a defective manner. Pairault wrote at the beginning of the century:
“In the sugar houses that produce them, these molasses are most often kept in large masonry basins dug in the ground and not covered. These molasses are thus exposed to all the dusts of the plant, moreover, it is frequent that rats (not to mention the many insects) fall into it and die there, and even more often than once for larger animals. Drawn from these basins, the molasses is introduced into boucauts (barrels of 500 liters), which are used for its transport and whose internal cleaning is almost always insufficient. On their arrival in the rhummeries of the cities, the barrels are rolled on dusty soil and brought by special men on depots formed by planks arranged in a clear way over a pit similar to that of the factory. The dust brought by men’s feet falls into molasses. Syrup spreads over the beams, forms a viscous coating thickened by dust, molds grow on the whole, but no one cares.”
At present, molasses are generally kept in steel tanks, in the form of vertical or horizontal cylinders, and whose capacity can vary from 100,000 to more than 1,000,000 liters.
In Java, certain factories, in order to make transport and handling easier, make solidified molasses. The molasses, diluted with water so as to mark about 70° Brix, is introduced after having been heated and skimmed in a vacuum box. It is concentrated until only about 6% of water remains. The contents of the box are then poured and received in bamboo baskets lined with mats, where it becomes a hard and brittle mass. The molasses prepared in this way often undergo compositional changes, which diminish its value as a distillery raw material. During preparation, relatively large losses of dry matter occur. Reducing sugars are partially transformed into non-organic sugar by polymerization, all the more so as the temperature is higher.
Even when stored under excellent conditions, during its conservation, molasses undergoes a progressive inversion of the sucrose and a decrease of the total sugars. If the product is concentrated normally (about 50% of total sugars) and is kept at a moderate temperature, close to 30°, this is low, but losses can in some cases reach high proportions.
Bastide (1), in Java, examining molasses on their reception and after 4-5 months and 9-10 months intervals, observed: a reduction in polarization and sucrose level (average 0.23% after 4 months and 0.80 after 10 months), a decrease in fermentable reducing sugars (0.58% after 4 months and 1.21% after 18 months) and an increase in non-fermentable reducers, which rose on average from 3.42% to 3.99 after 4 months and at 4.21% after 10 months. Molasses obtained by the various purification processes (defecation, sulphitation or carbonation) behaved in a practically similar manner.
(1) Arch. Suikerind. Ned Indie XXXVIII, 701, 1930.
Koffler (1), in Cuba, also found a progressive reversal of sucrose, the average rate of which increased from 37.75% to 34.48% at the end of the 7th month, a decrease in Brix (1%), a decrease in pH (from 5.92 to 5.23 after 7 months) and a very noticeable accentuation of the coloring. The glucose level did not increase in parallel with the inversion of sucrose, so that there was finally a loss in total sugars.
(1) Int. S. J. XL. 108; 1938
Browne followed, for 21 years, compositional variations in two molasses samples from Cuba. Here are the results obtained by the author for one of these molasses:

On the other hand, the rate of non-sugar organic molasses reached above, in 1927, 34.68% dry matter, while in the ordinary molasses of Cuba, it is only 12.38% on average. Amino acids had disappeared and the color had gradually become darker and darker.
In addition to this slow decrease in saccharin richness, much more abrupt deterioration has been reported, molasses undergoing spontaneous decomposition, with temperature rise and release of gas.
Prinsen-Geerligs describes the phenomenon, known as froth fermentation, as follows:
“During cooling of the low-purity cooked masses, the surface swells and sometimes dies, a blackish foam filtering through the crevices. This foam covers the entire surface, rising continuously and sometimes overflowing the tank. In the case of final molasses, which do not form a crystallized superficial crust, the whole mass begins to foam from the bottom, but the phenomenon also has the same appearance as cooked masses…”
The phenomenon is accompanied by a strong release of heat, the temperature being able to rise to 100°. The emission of gases, formed by a mixture of CO2, formic acid, acetic acid and acrid fumes of unknown composition, is sometimes violent enough to give rise to projections of matter. After a few days, the mass can be transformed, in part or even entirely, into a charcoal, hard and porous, resembling carbonized sugar.
Cases of this nature have been reported particularly in Egypt, affecting molasses stored in silos dug in the ground (Habif, Neuville). A sample of charred molasses, examined by Habif (1), had the following composition:
(1) Bull. Ass. Chim. LIV, 809. 1937.

Prinsen-Geerligs also cites examples of bursting of vacuum boxes, during the preparation of molasses solidified in Java, as a result of spontaneous decomposition during the concentration.
A more attenuated form of froth fermentation is characterized by a production of CO2 which not only produces scums on the surface, but also forms more or less deep pockets inside the molasses tanks. Sometimes granular deposits of a carbonaceous material similar to that indicated above are formed. In the case of syrups and table molasses kept in closed boxes, pressure exerted by carbon dioxide may be sufficient to cause bursting of the containers (Browne).
It has long been recognized that froth fermentation of cooked masses and molasses was due, not to the activity of microorganisms, but to purely chemical reactions.
Prinsen-Geerligs hypothesized that it was caused by the spontaneous decomposition of unstable products resulting from the action of lime on sugars, and more particularly on reducing sugars, during the clarification of juices. Browne attributed a special role to glucic acid, whose lime salt readily absorbs oxygen from the air by producing formic acid and other products, with a great deal of heat. In its crystalline form, this acid can partially sublimate by the action of heat, finally giving compounds of dark color, rich in carbon. In the presence of organic impurities, it decomposes spontaneously.
It has been suggested that the reaction between amino acids and reducing sugars, which gives rise to very unstable compounds (Maillard), may also be involved in the spontaneous decomposition of molasses. This phenomenon, however, would be secondary and limited to the onset of decomposition, according to Browne, transformation continuing to occur after the disappearance of amino acids.
Slow deterioration of molasses during storage is generally regarded as a slower form of frothy fermentation (Prinsen-Geerligs, Browne).
The main causes that favor the latter appear to be the high concentration of molasses and the high temperatures during manufacturing and storage. Neuville (2) reports, for example, that molasses with 93.35 Brix, obtained in a sugar factory in Egypt in 1938, underwent a particularly rapid decomposition, while others having the same origin, but Brix only reached 90°, stored without any alteration. In Java, it is considered that the temperature during manufacture of solidified molasses should not exceed 75°, otherwise there is a risk of spontaneous decomposition. According to Honig (3), if molasses stored at 30-35°C undergo a 2-3% reduction in fermentable sugars per year, a rise in temperature of 10° would be sufficient to quadruple the losses in certain cases. It is advisable, therefore, to cool the molasses before sending them to storage tanks and to paint them in white to reduce heat absorption.
(2) Proc. 6. Cong. Int. Soc. Sugar Cane Techn. 971, 1939.
(3) Proc. 5 Cong. Int. Soo, Sugar Sane Techn. 228, 1935.
In some cases, however, reduction in the saccharine richness of molasses, is sometimes accompanied by various modifications of flavor and odor (molasses, tart or sour, moldy, etc.), is due to the intervention of ferments. Ashby, in Jamaica, has found a wild yeast (Torula) that can ferment molasses at 90° Brix. Tempany found the intervention of bacteria and molds in the ripening molasses of the Lesser Antilles. Owen, Church, Hucker and Pedersen have encountered many yeasts, bacteria and molds in molasses, some of which attack sugars in relatively high concentrations.
Some molds (especially Aspergillus repens) are able to determine the inversion of sucrose in solutions of 65 and even 70° Brix (Owen, Van Der Bijl). On the other hand, bacteria would exert an appreciable action only at concentrations lower than 60° Brix. According to Owen (1), it would be the same of most Torula encountered in molasses and whose activity would be limited mainly to the destruction of invert sugars. This last author gives the following example of the modifications undergone by a first molasses from Cuba, diluted to 71° Brix and seeded with a Torula:
(1) Sugar XXXVIII, No 9, 22, 1943.

Under normal conditions of storage, losses resulting from the action of the various ferments are generally of little importance. Absorption on the surface of the molasses may be sufficient to allow the growth of mold and the consequent production of some oxalic and citric acids, but the molasses layer attacked, usually remains very thin and represents only a small part of the total volume.
It is quite different when the tanks remain open in the open air or consist of pits dug in the ground. In this case, as a result of the supply of rainwater, the dilution of the molasses may be such that the butyric bacteria are able to develop and reproduce, if the storage is prolonged, to significant amounts of butyric acid.
[I have heard an anecdote of some of these pits being overcome by flood waters and starting methane production very similar to an anaerobic digestor.]
To reduce losses of sugar caused by microorganisms and to prevent production of unpleasant aromatic substances that can then pass into the rum, it is important to concentrate molasses sufficiently (85-90° Brix) and to store them in closed tanks. Tubs, pipes and molasses tanks should be properly disinfected.
Value as fermentation material
The value of molasses as raw material for the production of rum is very variable and, in general, significantly lower than that of cane juice.
During manufacture and during subsequent storage of molasses, sugars undergo chemical or biochemical transformations, which make them less easily fermentable (carameliform substances, glutose) or lead to production of substances (formic, butyric, oxalic acids, etc.) constituting, at slightly high doses, yeast poisons.
On the other hand, the ash content, often very high, can hinder the functioning of yeasts, as found by various authors (Reindel and Frey (2), Arroyo, etc.). The equilibrium of mineral matter, dominated by potash and lime salts, which in some cases (cane from salty soil) is very rich in Na chloride, sometimes leaves much to be desired. There are certainly cases where molasses musts are not a physiologically balanced solution. (1)
(2) Z. Spiritusind. LVII, 237, 1934.
(1) A solution in which the toxic properties possessed by various salts, if they were alone, are neutralized by the presence of other antagonistic salts. The type of physiologically balanced solutions is seawater.
The most suitable molasses for rum production are those which have a specific weight and high levels of sugar, nitrogen and phosphoric acid, together with a small amount of ash and gums. The aroma is also of great importance, the presence of unpleasant smells and flavors affect the distillate. Fresh molasses always produce better rum than those that have been stored for a long time. Older molasses have a higher viscosity and often give a blackish scum, which is usually accompanied by a large decrease in alcohol yield.
Arroyo gives the following examples of molasses with different qualities from the point of view of rhum fermentation.

These molasses gave, in 3 fermentation tests at different concentrations, the following results:

According to Arroyo, a molasses can be considered excellent, when the ratio total sugars: ash is equal to or greater than 6.5/1: as good, when it is between 6.5/1 and 4.5/1, and as very mediocre, when it is less than 4.5/1. Nitrogen content is sufficient if the total N content is 4% of the molasses by weight; below, it is important to add Ammonium sulphate. As for phosphoric acid, its optimum dose is between 0.2 and 0.25% in P2O5 [Phosphorus pentoxide]). The P205: N ratio should be about 1/5; an excess of phosphoric acid relative to the nitrogen greatly hampers the smooth operation of the fermentation.
The richness of molasses in microorganisms is generally between 10 and 25% of that of cane juice. These microorganisms are represented by bacteria (mainly subtilis, mesentericus and butyricus groups), molds and yeasts (usually of the Torula type).
According to Church, the amount of bacteria in molasses at the centrifuge outlet can vary between 10 and 2,000 per gram, while fungal-mold spores reach at least 40,000 and in some cases up to 200,000. Owen found that in final molasses, the number of microorganisms is likely to reach a maximum of 100,000 per gram.
Some cane juice bacteria can withstand spores at the high temperatures of sugar manufacture and are then found in molasses, but most of the microorganisms encountered come from infections during sugar turbination, manipulation and especially molasses storage.
These microorganisms generally play only a minor role in the alcoholic fermentation of musts, the molasses dilution rate being large enough to reduce their concentration to a small number on the one hand, and acidification of musts hindering their development on the other hand.
To achieve good conditions for alcoholic fermentation, it is important to dilute molasses properly, to acidify and to seed the musts. Addition of nutrient salts, and in particular of ammoniacal salts, may be advantageous in certain cases, as well as the elimination of part of the ashes and materials (organic acids, gums, etc.) hindering the development of the yeast. Molasses presenting large variations in composition tests will be necessary in all cases to specify the best treatment to be applied.
Syrups.
Syrups are concentrated cane juice, usually after lime defecation.
We use this raw material, on a small scale for that matter, to obtain rums of special quality, devoid of the “vesouté” taste of cane juice eaux-de-vie and possessing a finer bouquet than molasses rums. In addition, when sugar prices are low, it is more advantageous to produce syrups for export and as a raw material for biochemical industries. This is what happened during the sugar crisis in Cuba, where large quantities of cane syrup was made, under the name of high test molasses, for shipping to the United States.
Preparation.
Vesou cooked. — Cooked vesou is cane juice which is boiled rapidly for a few minutes, so as to have a final density of about 1.100. During boiling, the juice is carefully de-scummed. This product is still sometimes prepared in the French West Indies, in order to obtain a special rum intended solely for the consumption of a few amateurs.
Gros syrup. — Formerly known as gros sirop, the syrupy liquid, dark in color, from the dripping of raw sugars obtained by evaporation of cane juice.
This evaporation was done in a battery of 5 boilers, arranged in line on a solid masonry and heated by open fire. The set was called the crew or battery. The dimensions of these boilers, generally cast iron and hemispherical, decreased from the first to the last. The first, called the grand one, placed at the most hearty part, was used to perform the defecation of the vesou. The juice, after the addition of a small quantity of lime or ashes, and after the removal of the foam, was transferred by means of a bucket with a long handle into the second basin, called the clean one [la propre]. A certain amount of foam was still produced in it, which was rejected in the grand one. Syrup coming from the clean one received in the third boiler, or flambeau, an additional quantity of lime, if its limpidity left something to be desired, then was brought into the 4th boiler or sirop to a determined concentration. The cooking was finished in the 5th boiler, or battery. “The name battery,” says P. Labat, “is due to the fact that one is obliged to shake sharply with a big skimmer, to prevent it from overflowing.”
[Battery syrup is mentioned elsewhere in the text and is no doubt some kind of evolution of this basic process. Haiti may use battery syrup.]

FIG. – Battery of P. Labat and refreshers (Martinique nineteenth century).
In the various sugar-producing countries there were various modifications of the above model, known in the French Antilles as the battery of P. Labat, although it was earlier than the arrival of this monk in the West Indies.
In the equipment of Jamaica, for example, the hemispherical boilers, usually 3 in number, were preceded by a shallow, rectangular vessel placed at a higher level than the boilers and serving as a defecator.
Concretor, which has been used in some countries, consisted of a long plate (about 110 m), with partitions starting alternately on each opposite side. The plate was slightly inclined on its axis, so that the juice, received at the front part, flowed in a continuous stream on a small thickness, to the end of the apparatus. Under the tray was a furnace, but unlike the arrangement of other batteries, it was the part receiving the raw juice which was arranged above the hearth. The syrup arriving at the bottom of the apparatus entered a rotating cylinder, heated by steam coming from the vesou. In some areas, the device was reduced to the tray without its accessories.
The mass-baked out of the battery was poured into very flat tanks (coolers), where it was allowed to cool, then in barrels opened from the top and whose lower bottom was pierced with holes. The barrels were placed on a slatted floor covering a tank, where the syrup from the dripping of the raw sugar flowed. The length of stay in the purgerie was 3-4 weeks.
When one wanted to obtain sucre terré, the cooked mass was introduced in forms, conical vases in unglazed earth, carrying at their extremity an orifice for the flow of syrup. These forms were placed next to each other, tip down. Once the syrup had been drained, the soil was put to the terrage, pouring on the sugar a layer of clayey earth, diluted in a thick slurry. Water that was in the clay slowly escaped and spread in the mass of sugar, bringing with it the viscous syrup that was there. The first layer of soil being entirely dry, it was replaced by a second and often by a third until the sugar was white enough.
The quantity of gros syrup obtained reached about 50% of the weight of the sugars, if these were simply subjected to purging, and up to 60% if one carried out the terrage. According to Itier (1), in 1844, in Cayenne, for every 100 kg of sugar, 50 kg of molasses were obtained in the case of virgin cane, 33 kg with the first offspring and 20 kg with the second offspring.
(1) Ann. Marit. Col. 1844, t. 3. 626.
Manufacture of raw sugar by the crew system has been abandoned in the West Indies since the beginning of the century. The few remaining batteries produce only syrup for culinary consumption. However, in South America, and particularly in the Brazilian countryside, there are still facilities for the preparation of brown sugar.
Battery syrup. — Battery syrup was formerly obtained by the crews. The juice was treated in the same way as for the manufacture of sugar. However concentration was pushed only to around 65-70° Brix, which allowed removing the last basin used for cooking syrup. [only used 4 of the 5 basins]
In some islands of the West Indies (Barbados), as well as in the United States (Louisiana, Florida), there are still a number of facilities of variable importance which prepare syrups by evaporation of cane juice in open boilers. However, the product obtained is generally intended for table consumption. On the other hand, the old “crews”, which required considerable fuel expenditure, have been modernized. Cylindrical or rectangular evaporators constructed of copper or galvanized sheet are usually heated by means of steam coils. Continuous evaporators consisting of a long tray with transverse partitions to allow juice to flow slowly by gravity from the inlet to the outlet of the apparatus, are widespread in the United States (Walton, Ventre and McCalip (2).
(2) Farm production of sugarcane sirup Farmers Bull. no 1874, 1941.
In distilleries of a certain importance producing syrup rum, as found in the French West Indies, the concentration of vesou is done in vacuum apparatus with multiple effect analogous in number and dimensions to those of the sugar factories. Concentration of the syrup can vary from 55 to 75 ° Brix.
The juice is usually defecated with lime (3), as follows. It is first added, in heaters equipped with a vertical vane agitator, the amount of milk of lime necessary to reach the point of neutrality. It is then brought to the temperature of 80-90° in a tubular heater, then to that of boiling in open defecation boilers, equipped with a coil fed with live steam and generally 3 in number to allow continuous work.
(3) Sulphitation of the juice, sometimes practiced in Louisiana for the preparation of table syrup, is not used in the manufacture of distillery syrup.
The clear juices from the defecators are filtered with Philippe filter or on very fine mesh flat filters before being sent to the evaporation chambers. As for turbid juices, they are passed to filter presses, then sent to clear juice filters, to be mixed with the previous ones.
Liming juice is not always practiced. In this case the clear liquid from the defecators is sent as such for evaporation, without passage to the Philippe filter: the turbid juices are treated with filter press. In other cases, in order to prevent sugar crystallization, which occurs when the syrup reaches a high concentration, a certain quantity of sulfuric acid or tartaric acid (1 gr per kg of sugar) is added, to determine inversion of a part of the sucrose.
Invert syrup. — The product known as high test molasses is prepared in Cuba by partially inverting lime-defecated syrups which are neutralized and then concentrated to the normal density of molasses (82-84° Brix).
Inverting is usually done with sulfuric acid (1 to 2.5 liters per 1,000 liters of syrup) at temperatures varying between 75 and 90°. This procedure has several disadvantages. The rise in temperature destroys a certain amount of sugar (levulose). On the other hand, if the neutralization precedes concentration, abundant encrustations are formed in the evaporators, and if it follows it is very difficult to obtain a suitable mixture of lime and syrup.
Inversion by means of invertase avoids these disadvantages. In the Guerrero process, juice from the last mills, which, because of its high nitrogen content, is suitable for yeast culture, is pasteurized, cooled and seeded with a special yeast, which contains about 10 times more invertase than ordinary baker’s yeast. The yeast obtained after passing through a contractive separator is used to continuously seed the flow of syrup from the evaporators. 4 volumes of yeast suspension (concentration 12% of the original yeast milk) are used per 100 volumes of syrup at 53° Brix. After 10 hours, 65% of the sucrose of the syrup is inverted.
Composition.
The composition of syrups is even more variable than that of molasses, because of the variety of preparation techniques.
Gros syrups, or molasses of brown sugar, which present little more than historical interest today, differed from the present sugar molasses by their greater richness in crystallizable sugar and their relative poverty in reducing sugars and minerals. Here are some analyzes of gros syrups produced in Martinique and Louisiana.

Hereinafter the composition of battery syrups of various origins:

As for inverted syrups from Cuba, they contain 18 to 30% sucrose (average 23%) and 30 to 60% reducing sugars (average 52%). It should be noted, by way of comparison, that blackstrap molasses from the same country average 35% sucrose and 20% reducers.
As fermentation raw material, syrups are characterized by their lack of nitrogen and mineral matter. Musts prepared from these products require, in order to ferment properly, to be supplemented with ammonia nitrogen and certain nutrients (phosphates), and sometimes to be acidified (syrups defecated with lime). Syrups, with the exception of gros syrups, are particularly poor for ferments, as shown in the following table which indicates the number of microorganisms existing per cc or by gr of material at different stages of sugar production (Owen Church, Kopeloff):

Syrup must therefore be seeded with a pied de cuve. If left to spontaneous seeding, the start of fermentation will be very slow.
Foams, washings, etc…
In addition to molasses, sugar factories produce various by-products (defecation scums, washings), which are generally mixed with the preceding batch, but which in some cases can be used for the composition of special musts. On the other hand, in order to produce certain types of rum, sugary juices are sometimes subjected to treatments, which profoundly modify quality. This is particularly the case in the production of Jamaican rum, where “acid” and “aroma” added to the musts influence the nature of the fermentation and bouquet of the eau-de-vie obtained.
Scums. [Hard to say if écumes should be translated at scums or foams.]
This name refers to mousses and deposits that occur during defecation of cane juice.
Scums were formerly of great importance as rhummerie raw material, at the time of the “sugar houses”, when one made brown sugar by the crew process. After establishment of the central factories, in some cases (in Jamaica and sometimes in the French West Indies), skimmings obtained in the open defecation boilers (1) were sent to the distillery. But most often they are subjected to a filtration to recover the sweet juice, which goes into sugar manufacture. At present, factories using the Petree-Dorr treatment process, which tends to become widespread in the West Indies, no longer produce defecation scum.
(1) When defecating juice is brought to a high temperature in the heaters and hot milk of lime added, which usually takes place in modern sugar factories, impurities fall for the most part at the bottom of defecation boilers, constituting “sludge” rather than “scums”. Formerly on the contrary, when juice was not de-aerated by preheating, but after cold liming it was gradually carried in the defecator around the boiling, most of the impurities rose to the surface.
Scums, to which was added washing water from the boilers and various residues of the sugar mill, were received in a special vat, where they were left to decant, the clear liquid being alone used for the composition of musts. The deposit was discarded, sometimes after washing with water, so as to recover the remaining sugar.
“Scums, out of the sugar factory,” Ducoeurjoly writes (1802), “will be deposited in a cistern, or in a vase large enough to contain all those removed from the sugar boilers, in 48 consecutive hours of work. They should be used only after this space of time, which is necessary to establish a beginning of fermentation, which drives out and rejects on the surface all the refuse with which they are charged; by this means we greatly diminish the daily care we give ourselves to skim the grappes. We must not hurry to use the foam, it is essential to wait until they are well cleaned and cleansed, by their own fermentation… The materials that are removed, by scumming the vase where the foam is deposited, are excellent food for horses, oxen and mules, they fatten them, but we must not give it to them too hot.”
Wray wrote on his side: “Scums comprising all the substances separated from cane juices during the defecation and evaporation operations, we thus have the thick crust of scum formed on the surface of the liquid in the clarifiers, the scum of the precipitators and evaporators and deposits formed at the bottom of these two series of vases. A portion of sweet liquor is mixed with the different scums and deposits, plus a large amount of water that was used to clean various vases and pipes. Later, each time the liquid is removed from the evaporators, as happens every Saturday night, a certain amount of what is called “soft liquor”, resulting from washing the boilers, etc., will join the scum. All this flows together in the distillery, mixes in the container with the scum, and takes the name of “residues”. The liquid part of these residues, when clarified, is withdrawn and takes the name of skimmings proper…”
“When the residues of sugar manufacture flow to the distillery, they are received in what is called the scum container, which usually holds 300 and 400 gallons. This vase is always lined with rolled lead or very thin copper; it is equipped with a tap to extract its contents. A container being full, the liquid is allowed time to clear. The thinned liquor, then properly named skimmings, is drawn into the tank to the mixture if there is one, or in the opposite case it goes directly into the fermentation tank.”
“All remaining impurities in the container are emptied into another container immediately below the first, where they are mixed with 100 or 200 gallons of water (hot if available) and well mixed, long enough to separate all the sugar. This work must continue until the sugar has been washed well, the liquid mass then remains some time at rest to clear again. If there is reason to fear fermentation or acidity, in the first case, a little sulfuric acid is added, by burning one or two sulphured matches, and in the second, a certain quantity of lime. The clarified liquid can be poured into the fermentation tank to help prepare the liquid to be distilled. I know that the washing of the deposits is rarely used, but it certainly should be, otherwise what these deposits contain of sweet matter would be discarded.”
Scums usually entered a certain proportion in the composition of musts: Wray indicates that of 2 skimmings for every 1 of molasses, as the best and most economical. Rarely, they were used alone. Rich in microorganisms and nitrogenous matter, they contain a relatively small amount of sugar, they fermented very quickly and easily underwent acetic acidification. Mixed with molasses, they facilitated the start of fermentation. Musts obtained from the scums alone gave a rum with pronounced taste and often of inferior quality (burnt taste).
Boname (1) gives the following composition for raw (not decanted) scums:
(1) Culture de la canne & la Guadeloupe, Paris, 1888.

The ashes, relatively rich in phosphoric acid, contain, on average, according to the same author:

Allan indicates as composition clarified scums used in Jamaica in the composition of the musts:

Acids, total and volatile, are evaluated as sulfuric acid as in the analyzes of the same author which we reproduce later.
In Jamaica, scums are allowed to sour for a few days before using them. For the production of high-flavored rums, they are sent in tanks half-filled with bagasse and cane straw, where they stay for 4 to 6 days. The acidity of ripe scums is already higher than it is proposed to get a more full-bodied rum.
This acidity is mainly due to the presence of fixed acids, of which lactic acid predominates and which represent 75 to 80% of the total acids. Since fresh scums have a neutral reaction, the acids originate during the microbial fermentations that develop during storage of the scums. Ashby has been able to isolate from them many bacteria, among which he has almost always found lactic acid bacterium, which forms rounded gelatinous masses in the liquid.
Scums obtained in the past had a higher saccharin content than those indicated above. It was usually considered that 60 liters of scum had the same sugar content as 10 liters of molasses. In some cases, however, it took up to 1 hl of scums to give the same amount of rum as 10 liters of molasses.
Washings.
Formerly referred to as washings stricto sensu, the product from the washing trays containing molasses. The latter being badly exhausted, a layer, several inches thick, of a blackish sugar, which was dissolved by means of hot water, was deposited at the bottom of the vats. The liquid obtained, marking about 32° Brix, was often used alone in the preparation of musts. It went into fermentation very quickly and gave a light rum with a finer bouquet than molasses rum.
In a broader sense, liquids from the washing of the various containers which have contained sweet substances (juice and syrup trays, defecation boilers, etc.) are called washings. These liquids are normally sent to the distillery, where they are combined with scums or mixed, during the composition of musts, with other raw materials.
Acid and aroma.
In the manufacture of grand-arôme Jamaican rum (german rum type) cane juice is used which has been the subject of a preliminary treatment to develop certain aromatic principles (acids, esters).
The “acid” (acid) is obtained by subjecting juice, previously defecated with heat and adding vinasse and sometimes a little scum, to the alcoholic fermentation. When this is almost complete, the liquid is directed into tanks half filled with bagasse and cane straw, where it is allowed to acidify. From time to time deposits of fermented musts are also sent to these tanks. From the first tank, the liquid is transferred to a second, then to a third. At the end of the latter the “acid” is good to be used in must composition. It then has the smell of sour beer.
The “arôme” (flavour) is prepared by fermenting cane juice with vinasse from the previous year (clear part), in tanks containing bagasse. It also usually contains a certain amount of the liquid from the muck hole, which is sent the deposits of vinasse and small waters, appliance washing water, bagasse and other plant residues.
We give below, according to Allan, the composition of the products obtained. These are particularly rich in fixed acids.

The bottoms, formed mainly by yeast cells mixed with loose bagasse, seem to play a particularly important role in the “maturation” of acid and aroma (1). Those obtained in the manufacture of german rum have, according to Allan, as composition:
(1) According to Cousins, esters and high-molecular-weight acids, which play an important role in the formation of the bouquet of high-flavored Jamaica rums, would come from the bacterial decomposition of dead yeast cells found in fermentation muck and vinasse muck.

Boname gives the following analysis of 3 bottoms from the fermentation of molasses in Mauritius (gr per liter):

Vinasse. [Dunder]
Vinasse, more commonly known as vidange [emptying or drainage] in the French West Indies and dunder in English countries, is the residue of the distillation of fermented musts.
Formerly, in the French West Indies, vinasse was received at its output from the distillatory apparatus in large surface masonry ponds, in order to facilitate cooling. In the important distilleries, there were the continuation of several basins, which the liquid crossed successively, so as to leave the last sufficiently cooled for use. This cooling in the open air has the disadvantage of allowing the invasion of vinasse by all kinds of bacteria and harmful germs. Also, today, in somewhat large installations, tubular condensers are usually used, which bring the temperature of the liquid to 30-35°. When water is lacking, cascade refrigerants are also frequently used. These consist of several stages (5 to 10) of fascines or slats, on which the vinasse flows, distributed at the top of the condenser by wooden gutters pierced with holes.
From the point of view of composition and properties, it is advisable to distinguish vesou vinasse from molasses vinasse.
The first is a yellowish, sparse liquid, usually between 1.010 and 1.020 density (3 to 50 Brix). Its acidity is quite variable, depending on the purity of the fermentations. It is generally between 3 and 5 gr (as sulfruic acid per liter). In the beginning of the century, when fermentations were longer and less pure, vesou vinasse acidity reached 6 to 7 grams per liter (Pairault) in the French West Indies. Dry matter, usually between 30 and 50 gr. per liter, contains on average 4 gr. ash rich in phosphoric acid and potash.
Molasses vinasse is dark brown in color and has a much higher density than vesou: 1.040 to 1.075 (about 10 to 19° Brix), but sometimes as high as 1.090 when it comes from concentrated musts used in manufacturing grand arôme rums. Its average acidity, in the case of pure fermentations, is 4 to 6 gr. per liter. But it reaches up to 30 gr. per liter, for grand arôme rum vinasse. Between these extremes, all intermediaries can exist. Pairault, at the beginning of the century, indicated as average acidity from molasses vinasses of the French West Indies 10-11 gr per liter. The volatile acidity (formed mainly by formic acid) varies between 10 and 40% of the total acidity. Dry matter (80 to 200 grams per liter) comprises 20 to 50 gr. per liter of ash rich in potash; 3 to 9 gr. nitrogenous material; varying quantities, but generally low (2 to 4 grams per liter), sugars having escaped fermentation and unfermentable reducers.
Boname (1) gives the following percentage composition for molasses vinasse from Mauritius:
(1) Ann. Sc. Agron. (2) II 215, 1896.

The mineral substances were composed as follows (in grams per liter):

According to Pairault, the proportion of nitrogenous matter is rather variable. When sulphate of ammonia is added to musts, this salt, if it is used in excess, is found partly in the vinasse and increases the total nitrogen content. On the other hand, during storage, there is a deposit of nitrogen-rich materials, so that the results are substantially different depending on whether one operates with cloudy or clarified vinasse. The above author has found, for various vinasse from Martinique, a content of 7 to 9 gr total nitrogen content per liter. Nitrogenous materials were distributed as follows in a clear and slightly old sample of vinasse (in grams per 1):

Nelson and Greenleaf found as a percentage basis of the dry matter of a cane molasses vinasse from a Baltimore distillery:

In addition to acetic and formic acids, they identified the following fixed acids:

Tricarballylic acid, CO2H – CH2 – CH (CO2H) – CH2 – CO2H, comes from reduction of aconitic acid during fermentation. Succinic acid and most of lactic acid are also products of molasses fermentation.
Allan indicates as vinasse composition used in Jamaica for various types of rum manufacture:

In the manufacture of certain types of rum, vinasse is stored from one season to the next. Some distillers, in order to prevent alteration of the vats when they are empty and dry, also fill them with vinasse: the latter being acid does not putrefy and the vats are well preserved.
After a while, vinasse is covered with a thick veil of mold. which prevents access to the air, and clarifies itself, constituting a yellowish liquid of bitter taste, often used in the preparation of musts for grand arôme rum. [Percival] Greg found that it contained a very aromatic acid, which smells of ethyl acetate and lemon oil. This body, which the author named fruity acid and found in Jamaican rum, appears only after the vinasse has already undergone acetic fermentation and the formed acetic acid has disappeared in part. It can be extracted with petroleum ether.
Finally, vinasse, if not used immediately, contains many microorganisms, including yeasts of the type Schizosaccharomyces (Pairault) and bacteria (Kayser).
Vinasse plays an important role in the fermentation of musts and the production of rum bouquet. It brings appreciable quantities of nitrogen and mineral matter, yeast foods, and especially of acids, which promote esterification, protect the yeast against the competition from microbes and ensure predominance of certain races of ferments (1). The regulating role of vinasse has been known for a long time. Wray already wrote in 1848: “It increases the density of the liquid, prevents the excessive violence of the fermentation, during which so much alcohol is lost, and holds the liquor at a comparatively low temperature, in a slow and moderate internal working state.”
(1) Fixed acids, on the other hand, would play a useful role in helping to fix certain basic products made by bacteria which, which passing through the distillate, would be detrimental to the quality of the rum (Allan).
It also intervenes, both by the aromatic principles that it brings and the influence it exerts on fermental flora, in the formation of the bouquet. Rums obtained by using high proportions of vinasse are less fine, but more full-bodied and more aromatic than the others.
Finally, as there is sometimes a certain amount of fermentable sugars as a result of defective fermentations or even alcohol, return of the vinasse in the manufacture can lead to fairly significant increases in yield.
In the beginning of the rhum industry, it was even avoided to push the alcohol exhaustion of the vinasses too far. “In order for them to have all the qualities demanded of them,” writes Ducœrjoly (1802), “the distillation of the little water must not be too much, for fear that it will strip them entirely of all their spirit parts, and do not impoverish them to the point of being no longer fit to be employed.”
Distillation was stopped when the alcoholic strength of the small waters [tails] fell around 34° G.L.
Dilution water.
The chemical and bacteriological composition of the water used in the preparation of musts influences the course of fermentation and the product quality obtained.
Mineral substances contained in the water, particularly the salts of soda, magnesia, lime and iron, increase the value of the nonsugar ratio: sugars, which, in the case of molasses musts, principally may be detrimental to the development and the diastatic activity of yeast. It is especially in the last stages of fermentation that the harmful action of these salts is felt. The typical fermentation mechanism can be more or less disturbed, with formation of secondary products, which modify the bouquet of the eau-de-vie. In addition, during distillation, they cause deposits on the trays and in the supply lines of the columns reducing performance and forcing frequent stops to clean the devices.
In principle, waters heavily loaded with mineral matter (calcareous, magnesian, ferruginous waters) are to be rejected. The same is true of seawater, which is however sometimes used and may in certain cases (musts low in mineral salts) (2) have a useful effect on fermentation (see Chapter V.).
(2) The use of seawater is reported by ancient authors. Ducœurjoly especially recommends it in the case of musts prepared without vinasse and scums, at the rate of 25 of sea water per 100 of ordinary water.
Water contaminated with organic matter, polluted by manure pits or in any other way is particularly harmful, as a result of the presence of decomposing materials (ammoniacal combinations, nitrites, nitrates, chlorine compounds, etc.) toxic to yeast and which can pass partly in the distillate.
Bacteria contained in the water, especially in those of pools and groundwater polluted with organic matter, cause secondary fermentations and give rise to volatile products of unpleasant smell or taste.
Water intended for the preparation of musts must therefore be as pure as possible. Unfortunately, industrialists are often forced to work with water of inferior and even frankly bad quality. In the French West Indies in particular, where the rum season usually coincides with the dry season, distillers frequently draw from the rivers at low water level, soiled by local residents, who come to wash their clothes.
Bettinger, however, did not find any significant differences in yield using, for must composition, both ordinary water and soapy water from the creek.
It is useful in some cases to correct the water intended for manufacturing. Those with high levels of nitrites, nitrates, chlorides or organic matter are not likely to be effectively treated. On the other hand, waters which contain carbonate of soda, potash, lime or magnesia are immediately improved by the addition of a quantity of dilute sulfuric acid, calculated so as to neutralize the alkalinity of these carbonates. The ferrous waters, which weaken yeast, can be treated by aerating them strongly by passing over beds of coarse gravel: the iron, which is mainly in the form of ferrous carbonate, precipitates in the state of oxide, which is then separated by sand filtration. Finally, fine sand filtration is suitable for murky waters.
Boiling is an effective method for ensuring bacteriological purity of water, but it is too expensive to be practical. The actual treatment method applicable in the fermentation industries is sterilization by ultraviolet rays or, better, by ozone.
2 thoughts on “Kervegant Chapter II Raw Materials of the Rhummerie”